AAV-mediated gene therapy in the guanylate cyclase (RetGC1/RetGC2) double knockout mouse model of Leber congenital amaurosis
- PMID: 23210611
- PMCID: PMC3581260
- DOI: 10.1089/hum.2012.193
AAV-mediated gene therapy in the guanylate cyclase (RetGC1/RetGC2) double knockout mouse model of Leber congenital amaurosis
Abstract
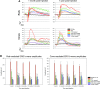
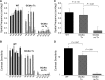
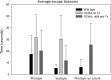
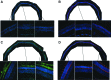
Mutations in GUCY2D are associated with recessive Leber congenital amaurosis-1 (LCA1). GUCY2D encodes photoreceptor-specific, retinal guanylate cyclase-1 (RetGC1). Reports of retinal degeneration in LCA1 are conflicting; some describe no obvious degeneration and others report loss of both rods and cones. Proof of concept studies in models representing the spectrum of phenotypes is warranted. We have previously demonstrated adeno-associated virus (AAV)-mediated RetGC1 is therapeutic in GC1ko mice, a model exhibiting loss of cones only. The purpose of this study was to characterize AAV-mediated gene therapy in the RetGC1/RetGC2 double knockout (GCdko) mouse, a model lacking rod and cone function and exhibiting progressive loss of both photoreceptor subclasses. Use of this model also allowed for the evaluation of the functional efficiency of transgenic RetGC1 isozyme. Subretinal delivery of AAV8(Y733F) vector containing the human rhodopsin kinase (hGRK1) promoter driving murine Gucy2e was performed in GCdko mice at various postnatal time points. Treatment resulted in restoration of rod and cone function at all treatment ages and preservation of retinal structure in GCdko mice treated as late as 7 weeks of age. Functional gains and structural preservation were stable for at least 1 year. Treatment also conferred cortical- and subcortical-based visually-guided behavior. Functional efficiency of transgenic RetGC1 was indistinguishable from that of endogenous isozyme in congenic wild-type (WT) mice. This study clearly demonstrates AAV-mediated RetGC1 expression restores function to and preserves structure of rod and cone photoreceptors in a degenerative model of retinal guanylate cyclase deficiency, further supporting development of an AAV-based vector for treatment of LCA1.
Figures


References
-
- Auricchio A. Fighting blindness with adeno-associated virus serotype 8. Hum. Gene Ther. 2011;22:1169–1170. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- EY08571/EY/NEI NIH HHS/United States
- RR025777/RR/NCRR NIH HHS/United States
- EY13729/EY/NEI NIH HHS/United States
- R01 AG014979/AG/NIA NIH HHS/United States
- R37 AG036800/AG/NIA NIH HHS/United States
- EY11522/EY/NEI NIH HHS/United States
- P30 AG028740/AG/NIA NIH HHS/United States
- EY11123/EY/NEI NIH HHS/United States
- U10 EY013729/EY/NEI NIH HHS/United States
- P30 EY008571/EY/NEI NIH HHS/United States
- P30-EY021721/EY/NEI NIH HHS/United States
- R01 EY011123/EY/NEI NIH HHS/United States
- R01 EY008123/EY/NEI NIH HHS/United States
- P30 EY021721/EY/NEI NIH HHS/United States
- P30 EY014800/EY/NEI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

