Effects of Fe(II)/H2O2 oxidation on ubiquitin conformers measured by ion mobility-mass spectrometry
- PMID: 23211023
- PMCID: PMC3552375
- DOI: 10.1021/jp3099544
Effects of Fe(II)/H2O2 oxidation on ubiquitin conformers measured by ion mobility-mass spectrometry
Abstract
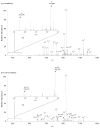
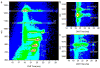
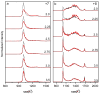
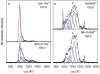
Oxidative modifications can have significant effects on protein structure in solution. Here, the structures and stabilities of oxidized ubiquitin ions electrosprayed from an aqueous solution (pH 2) are studied by ion mobility spectrometry-mass spectrometry (IMS-MS). IMS-MS has proven to be a valuable technique to assess gas phase and in many cases, solution structures. Herein, in vitro oxidation is performed by Fenton chemistry with Fe(II)/hydrogen peroxide. Most molecules in solution remain unmodified, whereas ∼20% of the population belongs to an M+16 Da oxidized species. Ions of low charge states (+7 and +8) show substantial variance in collision cross section distributions between unmodified and oxidized species. Novel and previously reported gaussian conformers are used to model cross section distributions for +7 and +8 oxidized ubiquitin ions, respectively, in order to correlate variances in observed gas-phase distributions to changes in populations of solution states. Based on gaussian modeling, oxidized ions of charge state +7 have an A-state conformation which is more populated for oxidized relative to unmodified ions. Oxidized ubiquitin ions of charge state +8 have a distribution of conformers arising from native-state ubiquitin and higher intensities of A- and U-state conformers relative to unmodified ions. This work provides evidence that incorporation of a single oxygen atom to ubiquitin leads to destabilization of the native state in an acidic solution (pH ∼2) and to unfolding of gas-phase compact structures.
Figures


Similar articles
-
Solution dependence of the collisional activation of ubiquitin [M + 7H](7+) ions.J Am Soc Mass Spectrom. 2014 Dec;25(12):2000-8. doi: 10.1007/s13361-014-0834-y. J Am Soc Mass Spectrom. 2014. PMID: 24658799 Free PMC article.
-
Evidence for two new solution states of ubiquitin by IMS-MS analysis.J Phys Chem B. 2014 Apr 3;118(13):3498-506. doi: 10.1021/jp4097327. Epub 2014 Mar 24. J Phys Chem B. 2014. PMID: 24625065 Free PMC article.
-
Comparison of gaseous ubiquitin ion structures obtained from a solid and solution matrix using ion mobility spectrometry/mass spectrometry.Rapid Commun Mass Spectrom. 2021 Jan;35 Suppl 1:e8793. doi: 10.1002/rcm.8793. Epub 2020 Aug 20. Rapid Commun Mass Spectrom. 2021. PMID: 32220130
-
Ion Mobility and Gas-Phase Covalent Labeling Study of the Structure and Reactivity of Gaseous Ubiquitin Ions Electrosprayed from Aqueous and Denaturing Solutions.J Am Soc Mass Spectrom. 2020 May 6;31(5):1037-1046. doi: 10.1021/jasms.9b00138. Epub 2020 Apr 16. J Am Soc Mass Spectrom. 2020. PMID: 32255627 Free PMC article.
-
THE IMS PARADOX: A PERSPECTIVE ON STRUCTURAL ION MOBILITY-MASS SPECTROMETRY.Mass Spectrom Rev. 2021 May;40(3):280-305. doi: 10.1002/mas.21642. Epub 2020 Jul 1. Mass Spectrom Rev. 2021. PMID: 32608033 Free PMC article. Review.
Cited by
-
Multiple proteases to localize oxidation sites.PLoS One. 2015 Mar 16;10(3):e0116606. doi: 10.1371/journal.pone.0116606. eCollection 2015. PLoS One. 2015. PMID: 25775238 Free PMC article.
-
Solution dependence of the collisional activation of ubiquitin [M + 7H](7+) ions.J Am Soc Mass Spectrom. 2014 Dec;25(12):2000-8. doi: 10.1007/s13361-014-0834-y. J Am Soc Mass Spectrom. 2014. PMID: 24658799 Free PMC article.
-
Gridless overtone mobility spectrometry.Anal Chem. 2013 Nov 5;85(21):10174-9. doi: 10.1021/ac401568r. Epub 2013 Oct 14. Anal Chem. 2013. PMID: 24125033 Free PMC article.
-
Developments in Trapped Ion Mobility Mass Spectrometry to Probe the Early Stages of Peptide Aggregation.J Am Soc Mass Spectrom. 2023 Feb 1;34(2):193-204. doi: 10.1021/jasms.2c00253. Epub 2023 Jan 12. J Am Soc Mass Spectrom. 2023. PMID: 36633834 Free PMC article.
-
Evidence for two new solution states of ubiquitin by IMS-MS analysis.J Phys Chem B. 2014 Apr 3;118(13):3498-506. doi: 10.1021/jp4097327. Epub 2014 Mar 24. J Phys Chem B. 2014. PMID: 24625065 Free PMC article.
References
-
- Finkel T, Holbrook NJ. Nature. 2000;408:239–247. - PubMed
-
- Valko M, Leibfritz D, Moncol J, Cronin MT, Mazur M, Telser J. Int J Biochem Cell Biol. 2007;39:44–84. - PubMed
-
- Davies MJ. Biochim Biophys Acta. 2005;1703:93–109. - PubMed
-
- von Zglinicki T. Trends Biochem Sci. 2002;27:339–344. - PubMed
-
- Halliwell B. J Neurochem. 2006;97:1634–1658. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

