Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin
- PMID: 23212718
- PMCID: PMC3552520
- DOI: 10.1161/CIRCULATIONAHA.112.117283
Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin
Abstract
Background: Low dose aspirin reduces the secondary incidence of myocardial infarction and stroke. Drug resistance to aspirin might result in treatment failure. Despite this concern, no clear definition of aspirin resistance has emerged, and estimates of its incidence have varied remarkably. We aimed to determine the commonality of a mechanistically consistent, stable, and specific phenotype of true pharmacological resistance to aspirin-such as might be explained by genetic causes.
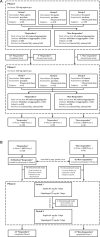
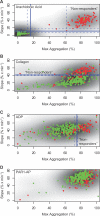
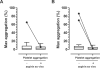
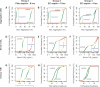
Methods and results: Healthy volunteers (n=400) were screened for their response to a single oral dose of 325-mg immediate release or enteric coated aspirin. Response parameters reflected the activity of the molecular target of aspirin, cyclooxygenase-1. Individuals who appeared aspirin resistant on 1 occasion underwent repeat testing, and if still resistant were exposed to low-dose enteric coated aspirin (81 mg) and clopidogrel (75 mg) for 1 week each. Variable absorption caused a high frequency of apparent resistance to a single dose of 325-mg enteric coated aspirin (up to 49%) but not to immediate release aspirin (0%). All individuals responded to aspirin on repeated exposure, extension of the postdosing interval, or addition of aspirin to their platelets ex vivo.
Conclusions: Pharmacological resistance to aspirin is rare; this study failed to identify a single case of true drug resistance. Pseudoresistance, reflecting delayed and reduced drug absorption, complicates enteric coated but not immediate release aspirin administration.
Clinical trial registration: URL: http://www.clinicaltrials.gov. Unique identifier: NCT00948987.
Figures
Comment in
-
Letter by Dillinger et al regarding article, "Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin".Circulation. 2013 Sep 17;128(12):e189. doi: 10.1161/CIRCULATIONAHA.112.001179. Circulation. 2013. PMID: 24043151 No abstract available.
-
Letter by Wallace and Chan regarding article, "Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin".Circulation. 2013 Sep 17;128(12):e190. doi: 10.1161/CIRCULATIONAHA.113.001433. Circulation. 2013. PMID: 24043152 No abstract available.
-
Response to letters regarding article, "Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin".Circulation. 2013 Sep 17;128(12):e191. doi: 10.1161/CIRCULATIONAHA.113.004750. Circulation. 2013. PMID: 24043153 No abstract available.
Similar articles
-
Response to letters regarding article, "Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin".Circulation. 2013 Sep 17;128(12):e191. doi: 10.1161/CIRCULATIONAHA.113.004750. Circulation. 2013. PMID: 24043153 No abstract available.
-
Letter by Wallace and Chan regarding article, "Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin".Circulation. 2013 Sep 17;128(12):e190. doi: 10.1161/CIRCULATIONAHA.113.001433. Circulation. 2013. PMID: 24043152 No abstract available.
-
Letter by Dillinger et al regarding article, "Drug resistance and pseudoresistance: an unintended consequence of enteric coating aspirin".Circulation. 2013 Sep 17;128(12):e189. doi: 10.1161/CIRCULATIONAHA.112.001179. Circulation. 2013. PMID: 24043151 No abstract available.
-
Aspirin in patients with coronary artery disease: is it simply irresistible?J Thromb Thrombolysis. 2001 Apr;11(2):117-26. doi: 10.1023/a:1011220615447. J Thromb Thrombolysis. 2001. PMID: 11406726 Review. No abstract available.
-
Terms and conditions: semantic complexity and aspirin resistance.Circulation. 2004 Sep 21;110(12):1706-8. doi: 10.1161/01.CIR.0000142056.69970.DB. Circulation. 2004. PMID: 15381661 Review. No abstract available.
Cited by
-
Personalized Dual Antiplatelet Therapy in Acute Coronary Syndromes: Striking a Balance Between Bleeding and Thrombosis.Curr Cardiol Rep. 2023 Jul;25(7):693-710. doi: 10.1007/s11886-023-01892-9. Epub 2023 Jun 1. Curr Cardiol Rep. 2023. PMID: 37261665 Free PMC article. Review.
-
Aspirin hydrolysis in plasma is a variable function of butyrylcholinesterase and platelet-activating factor acetylhydrolase 1b2 (PAFAH1b2).J Biol Chem. 2013 Apr 26;288(17):11940-8. doi: 10.1074/jbc.M112.427674. Epub 2013 Mar 18. J Biol Chem. 2013. PMID: 23508960 Free PMC article.
-
Impact of genetic polymorphisms related to clopidogrel or acetylsalicylic acid pharmacology on clinical outcome in Chinese patients with symptomatic extracranial or intracranial stenosis.Eur J Clin Pharmacol. 2016 Oct;72(10):1195-1204. doi: 10.1007/s00228-016-2094-1. Epub 2016 Jul 23. Eur J Clin Pharmacol. 2016. PMID: 27450232 Clinical Trial.
-
Platelet function testing and tailored antiplatelet therapy.J Cardiovasc Transl Res. 2013 Jun;6(3):316-28. doi: 10.1007/s12265-013-9458-z. Epub 2013 Mar 30. J Cardiovasc Transl Res. 2013. PMID: 23543615 Review.
-
Prerace aspirin to protect susceptible runners from cardiac arrest during marathons: is opportunity knocking?Open Heart. 2015 Jul 2;2(1):e000102. doi: 10.1136/openhrt-2014-000102. eCollection 2015. Open Heart. 2015. PMID: 26167288 Free PMC article. Review.
References
-
- Patrono C, Garcia Rodriguez LA, Landolfi R, Baigent C. Low-dose aspirin for the prevention of atherothrombosis. N Engl J Med. 2005;353:2373–2383. - PubMed
-
- Baigent C, Blackwell L, Collins R, Emberson J, Godwin J, Peto R, Buring J, Hennekens C, Kearney P, Meade T, Patrono C, Roncaglioni MC, Zanchetti A. Aspirin in the primary and secondary prevention of vascular disease: Collaborative meta-analysis of individual participant data from randomised trials. Lancet. 2009;373:1849–1860. - PMC - PubMed
-
- Funk CD, Funk LB, Kennedy ME, Pong AS, FitzGerald GA. Human platelet/erythroleukemia cell prostaglandin g/h synthase: Cdna cloning, expression, and gene chromosomal assignment. Faseb J. 1991;5:2304–2312. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

