Activity-dependent callosal axon projections in neonatal mouse cerebral cortex
- PMID: 23213574
- PMCID: PMC3507157
- DOI: 10.1155/2012/797295
Activity-dependent callosal axon projections in neonatal mouse cerebral cortex
Abstract
Callosal axon projections are among the major long-range axonal projections in the mammalian brain. They are formed during the prenatal and early postnatal periods in the mouse, and their development relies on both activity-independent and -dependent mechanisms. In this paper, we review recent findings about the roles of neuronal activity in callosal axon projections. In addition to the well-documented role of sensory-driven neuronal activity, recent studies using in utero electroporation demonstrated an essential role of spontaneous neuronal activity generated in neonatal cortical circuits. Both presynaptic and postsynaptic neuronal activities are critically involved in the axon development. Studies have begun to reveal intracellular signaling pathway which works downstream of neuronal activity. We also review several distinct patterns of neuronal activity observed in the developing cerebral cortex, which might play roles in activity-dependent circuit construction. Such neuronal activity during the neonatal period can be disrupted by genetic factors, such as mutations in ion channels. It has been speculated that abnormal activity caused by such factors may affect activity-dependent circuit construction, leading to some developmental disorders. We discuss a possibility that genetic mutation in ion channels may impair callosal axon projections through an activity-dependent mechanism.
Figures
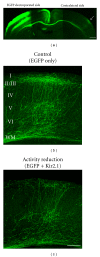


References
-
- Hubel D, Wiesel T. Cortical and callosal connections concerned with the vertical meridian of visual fields in the cat. Journal of Neurophysiology. 1967;30(6):1561–1573. - PubMed
-
- Shatz CJ. Anatomy of interhemispheric connections in the visual system of Boston Siamese and ordinary cats. Journal of Comparative Neurology. 1977;173(3):497–518. - PubMed
-
- Olavarria J, Montero VM. Relation of callosal and striate-extrastriate cortical connections in the rat: morphological definition of extrastriate visual areas. Experimental Brain Research. 1984;54(2):240–252. - PubMed

