Evaluation of osteoconductive scaffolds in the canine femoral multi-defect model
- PMID: 23215980
- PMCID: PMC3568967
- DOI: 10.1089/ten.TEA.2012.0289
Evaluation of osteoconductive scaffolds in the canine femoral multi-defect model
Abstract
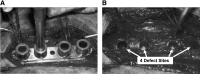
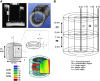
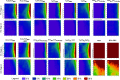
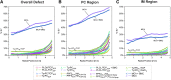
Treatment of large segmental bone defects remains an unsolved clinical challenge, despite a wide array of existing bone graft materials. This project was designed to rapidly assess and compare promising biodegradable osteoconductive scaffolds for use in the systematic development of new bone regeneration methodologies that combine scaffolds, sources of osteogenic cells, and bioactive scaffold modifications. Promising biomaterials and scaffold fabrication methods were identified in laboratories at Rutgers, MIT, Integra Life Sciences, and Mayo Clinic. Scaffolds were fabricated from various materials, including poly(L-lactide-co-glycolide) (PLGA), poly(L-lactide-co-ɛ-caprolactone) (PLCL), tyrosine-derived polycarbonate (TyrPC), and poly(propylene fumarate) (PPF). Highly porous three-dimensional (3D) scaffolds were fabricated by 3D printing, laser stereolithography, or solvent casting followed by porogen leaching. The canine femoral multi-defect model was used to systematically compare scaffold performance and enable selection of the most promising substrate(s) on which to add cell sourcing options and bioactive surface modifications. Mineralized cancellous allograft (MCA) was used to provide a comparative reference to the current clinical standard for osteoconductive scaffolds. Percent bone volume within the defect was assessed 4 weeks after implantation using both MicroCT and limited histomorphometry. Bone formed at the periphery of all scaffolds with varying levels of radial ingrowth. MCA produced a rapid and advanced stage of bone formation and remodeling throughout the defect in 4 weeks, greatly exceeding the performance of all polymer scaffolds. Two scaffold constructs, TyrPC(PL)/TCP and PPF4(SLA)/HA(PLGA) (Dip), proved to be significantly better than alternative PLGA and PLCL scaffolds, justifying further development. MCA remains the current standard for osteoconductive scaffolds.
Figures





References
-
- Owens B.D., et al. Characterization of extremity wounds in operation Iraqi freedom and operation enduring freedom. J Orthop Trauma. 2007;21:254. - PubMed
-
- Muschler G.F. Nakamoto C. Griffith L.G. Engineering principles of clinical cell-based tissue engineering. J Bone Joint Surg Am. 2004;86-A:1541. - PubMed
-
- Griffith L.G. Emerging design principles in biomaterials and scaffolds for tissue engineering. Ann N Y Acad Sci. 2002;961:83. - PubMed
-
- Griffith L.G. Naughton G.K. Tissue engineering—current challenges and expanding opportunities. Science. 2002;295:1009. - PubMed
-
- Hubbell J.A. Bioactive biomaterials. Curr Opinion Biotech. 1999;10:123. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

