Therapeutic effects of deleting cancer-associated fibroblasts in cholangiocarcinoma
- PMID: 23221385
- PMCID: PMC3549008
- DOI: 10.1158/0008-5472.CAN-12-2130
Therapeutic effects of deleting cancer-associated fibroblasts in cholangiocarcinoma
Abstract
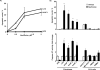
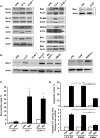
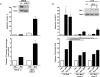
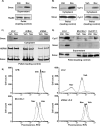
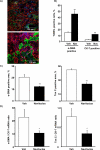
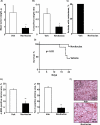
Cancer-associated fibroblasts (CAF) are abundant in the stroma of desmoplastic cancers where they promote tumor progression. CAFs are "activated" and as such may be uniquely susceptible to apoptosis. Using cholangiocarcinoma as a desmoplastic tumor model, we investigated the sensitivity of liver CAFs to the cytotoxic drug navitoclax, a BH3 mimetic. Navitoclax induced apoptosis in CAF and in myofibroblastic human hepatic stellate cells but lacked similar effects in quiescent fibroblasts or cholangiocarcinoma cells. Unlike cholangiocarcinoma cells, neither CAF nor quiescent fibroblasts expressed Mcl-1, a known resistance factor for navitoclax cytotoxicity. Explaining this paradox, we found that mitochondria isolated from CAFs or cells treated with navitoclax both released the apoptogenic factors Smac and cytochrome c, suggesting that they are primed for cell death. Such death priming in CAFs appeared to be due, in part, to upregulation of the proapoptotic protein Bax. Short hairpin RNA-mediated attenuation of Bax repressed navitoclax-mediated mitochondrial dysfunction, release of apoptogenic factors, and apoptotic cell death. In a syngeneic rat model of cholangiocarcinoma, navitoclax treatment triggered CAF apoptosis, diminishing expression of the desmoplastic extracellular matrix protein tenascin C, suppressing tumor outgrowth, and improving host survival. Together, our findings argue that navitoclax may be useful for destroying CAFs in the tumor microenvironment as a general strategy to attack solid tumors.
Figures
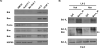
References
-
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74. - PubMed
-
- Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6:392–401. - PubMed
-
- Sirica AE. The role of cancer-associated myofibroblasts in intrahepatic cholangiocarcinoma. Nat Rev Gastro Hepat. 2011;9:44–54. - PubMed
-
- Rasanen K, Vaheri A. Activation of fibroblasts in cancer stroma. Exp Cell Res. 2010;316:2713–22. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

