N-geranyl cyclopropyl-carboximide modulates salty and umami taste in humans and animal models
- PMID: 23221408
- PMCID: PMC3569124
- DOI: 10.1152/jn.00124.2012
N-geranyl cyclopropyl-carboximide modulates salty and umami taste in humans and animal models
Abstract
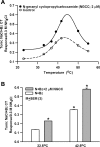
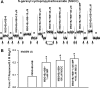
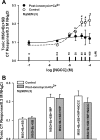
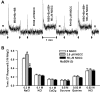
Effects of N-geranyl cyclopropyl-carboxamide (NGCC) and four structurally related compounds (N-cyclopropyl E2,Z6-nonadienamide, N-geranyl isobutanamide, N-geranyl 2-methylbutanamide, and allyl N-geranyl carbamate) were evaluated on the chorda tympani (CT) nerve response to NaCl and monosodium glutamate (MSG) in rats and wild-type (WT) and TRPV1 knockout (KO) mice and on human salty and umami taste intensity. NGCC enhanced the rat CT response to 100 mM NaCl + 5 μM benzamil (Bz; an epithelial Na(+) channel blocker) between 1 and 2.5 μM and inhibited it above 5 μM. N-(3-methoxyphenyl)-4-chlorocinnamid (SB-366791, a TRPV1t blocker) inhibited the NaCl+Bz CT response in the absence and presence of NGCC. Unlike the WT mice, no NaCl+Bz CT response was observed in TRPV1 KO mice in the absence or presence of NGCC. NGCC enhanced human salt taste intensity of fish soup stock containing 60 mM NaCl at 5 and 10 μM and decreased it at 25 μM. Rat CT responses to NaCl+Bz and human salt sensory perception were not affected by the above four structurally related compounds. Above 10 μM, NGCC increased the CT response to MSG+Bz+SB-366791 and maximally enhanced the response between 40 and 60 μM. Increasing taste cell Ca(2+) inhibited the NGCC-induced increase but not the inosine monophosphate-induced increase in glutamate response. Addition of 45 μM NGCC to chicken broth containing 60 mM sodium enhanced the human umami taste intensity. Thus, depending upon its concentration, NGCC modulates salt taste by interacting with the putative TRPV1t-dependent salt taste receptor and umami taste by interacting with a Ca(2+)-dependent transduction pathway.
Figures



Similar articles
-
TRPM5-dependent amiloride- and benzamil-insensitive NaCl chorda tympani taste nerve response.Am J Physiol Gastrointest Liver Physiol. 2013 Jul 1;305(1):G106-17. doi: 10.1152/ajpgi.00053.2013. Epub 2013 May 2. Am J Physiol Gastrointest Liver Physiol. 2013. PMID: 23639808 Free PMC article.
-
Regulation of the putative TRPV1t salt taste receptor by phosphatidylinositol 4,5-bisphosphate.J Neurophysiol. 2010 Mar;103(3):1337-49. doi: 10.1152/jn.00883.2009. Epub 2009 Dec 23. J Neurophysiol. 2010. PMID: 20032236 Free PMC article.
-
Effect of Maillard reacted peptides on human salt taste and the amiloride-insensitive salt taste receptor (TRPV1t).Chem Senses. 2008 Sep;33(7):665-80. doi: 10.1093/chemse/bjn033. Epub 2008 Jul 5. Chem Senses. 2008. PMID: 18603652 Free PMC article.
-
Differential Effect of TRPV1 Modulators on Neural and Behavioral Responses to Taste Stimuli.Nutrients. 2024 Nov 12;16(22):3858. doi: 10.3390/nu16223858. Nutrients. 2024. PMID: 39599644 Free PMC article. Review.
-
Responses to umami substances in taste bud cells innervated by the chorda tympani and glossopharyngeal nerves.J Nutr. 2000 Apr;130(4S Suppl):950S-3S. doi: 10.1093/jn/130.4.950S. J Nutr. 2000. PMID: 10736359 Review.
Cited by
-
TRPM5-dependent amiloride- and benzamil-insensitive NaCl chorda tympani taste nerve response.Am J Physiol Gastrointest Liver Physiol. 2013 Jul 1;305(1):G106-17. doi: 10.1152/ajpgi.00053.2013. Epub 2013 May 2. Am J Physiol Gastrointest Liver Physiol. 2013. PMID: 23639808 Free PMC article.
-
Interactions between Chemesthesis and Taste: Role of TRPA1 and TRPV1.Int J Mol Sci. 2021 Mar 25;22(7):3360. doi: 10.3390/ijms22073360. Int J Mol Sci. 2021. PMID: 33806052 Free PMC article. Review.
-
Kokumi Taste Active Peptides Modulate Salt and Umami Taste.Nutrients. 2020 Apr 24;12(4):1198. doi: 10.3390/nu12041198. Nutrients. 2020. PMID: 32344605 Free PMC article.
-
Pharmacology of the Umami Taste Receptor.Handb Exp Pharmacol. 2022;275:109-136. doi: 10.1007/164_2021_439. Handb Exp Pharmacol. 2022. PMID: 33580387
-
Selective activation of hTRPV1 by N-geranyl cyclopropylcarboxamide, an amiloride-insensitive salt taste enhancer.PLoS One. 2014 Feb 20;9(2):e89062. doi: 10.1371/journal.pone.0089062. eCollection 2014. PLoS One. 2014. PMID: 24586504 Free PMC article.
References
-
- Chaudhari N, Landin AM, Roper SD. A metabotropic glutamate receptor variant functions as a taste receptor. Nat Neurosci 3: 113–119, 2000 - PubMed
-
- Coleman J, Williams A, Phan TH, Mummalaneni S, Melone P, Ren ZJ, Zhou H, Mahavadi S, Murthy KS, Katsumata T, DeSimone JA, Lyall V. Strain differences in the neural, behavioral, and molecular correlates of sweet and salty taste in naive, ethanol- and sucrose-exposed P and NP rats. J Neurophysiol 106: 2606–2621, 2011 - PMC - PubMed
-
- Damak S, Rong M, Yasumatsu K, Kokrashvili Z, Pérez CA, Shigemura N, Yoshida R, Mosinger B, Jr, Glendinning JI, Ninomiya Y, Margolskee RF. Trpm5 null mice respond to bitter, sweet, and umami compounds. Chem Senses 31: 253–264, 2006 - PubMed
-
- Davis JB, Smart D, Gunthorpe MJ. The vanilloid receptor and vanilloid receptor-like genes: a hot topic getting hotter. Celltransmissions 18: 3–9, 2002
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

