Integrin α3β1 regulates tumor cell responses to stromal cells and can function to suppress prostate cancer metastatic colonization
- PMID: 23224938
- PMCID: PMC3604149
- DOI: 10.1007/s10585-012-9558-1
Integrin α3β1 regulates tumor cell responses to stromal cells and can function to suppress prostate cancer metastatic colonization
Abstract
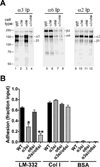
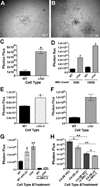
Integrin α3β1 promotes tumor cell adhesion, migration, and invasion on laminin isoforms, and several clinical studies have indicated a correlation between increased tumoral α3β1 integrin expression and tumor progression, metastasis, and poor patient outcomes. However, several other clinical and experimental studies have suggested that α3β1 can possess anti-metastatic activity in certain settings. To help define the range of α3β1 functions in tumor cells in vivo, we used RNAi to silence the α3 integrin subunit in an aggressive, in vivo-passaged subline of PC-3 prostate carcinoma cells. Loss of α3 integrin impaired adhesion and proliferation on the α3β1 integrin ligand, laminin-332 in vitro. Despite these deficits in vitro, the α3-silenced cells were significantly more aggressive in a lung colonization model in vivo, with a substantially increased rate of tumor growth that significantly reduced survival. In contrast, silencing the related α6 integrin subunit delayed metastatic growth in vivo. The increased colonization of α3-silenced tumor cells in vivo was recapitulated in 3D collagen co-cultures with lung fibroblasts or pre-osteoblast-like cells, where α3-silenced cells showed dramatically enhanced growth. The increased response of α3-silenced tumor cells to stromal cells in co-culture could be reproduced by fibroblast conditioned medium, which contains one or more heparin-binding factors that selectively favor the growth of α3-silenced cells. Our new data suggest a scenario in which α3β1 regulates tumor-host interactions within the metastatic tumor microenvironment to limit growth, providing some of the first direct evidence that specific loss of α3 function in tumor cells can have pro-metastatic consequences in vivo.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

