Injectable bone tissue engineering using expanded mesenchymal stem cells
- PMID: 23225744
- PMCID: PMC4043220
- DOI: 10.1002/stem.1300
Injectable bone tissue engineering using expanded mesenchymal stem cells
Abstract
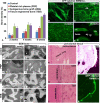
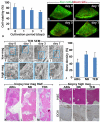
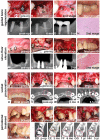
Patients suffering from bone defects are often treated with autologous bone transplants, but this therapy can cause many complications. New approaches are therefore needed to improve treatment for bone defects, and stem cell therapy presents an exciting alternative approach. Although extensive evidence from basic studies using stem cells has been reported, few clinical applications using stem cells for bone tissue engineering have been developed. We investigated whether injectable tissue-engineered bone (TEB) composed of mesenchymal stem cells (MSCs) and platelet-rich plasma was able to regenerate functional bone in alveolar deficiencies. We performed these studies in animals and subsequently carried out large-scale clinical studies in patients with long-term follow-up; these showed good bone formation using minimally invasive MSC transplantation. All patients exhibited significantly improved bone volume with no side effects. Newly formed bone areas at 3 months were significantly increased over the preoperation baseline (p < .001) and reached levels equivalent to that of native bone. No significant bone resorption occurred during long-term follow-up. Injectable TEB restored masticatory function in patients. This novel clinical approach represents an effective therapeutic utilization of bone tissue engineering.
Copyright © 2012 AlphaMed Press.
Figures
References
-
- Turkyilmaz I, Company AM, McGlumphy EA. Should edentulous patients be constrained to removable complete dentures? The use of dental implants to improve the quality of life for edentulous patients. Gerodontology. 2010;27:3–10. - PubMed
-
- Wang S, Zhao J, Zhang W, et al. Maintenance of phenotype and function of cryopreserved bone-derived cells. Biomaterials. 2011;32:3739–3749. - PubMed
-
- Joshi A, Kostakis GC. An investigation of post-operative morbidity following iliac crest graft harvesting. Br Dent J. 2004;196:167–171. - PubMed
-
- Damien CJ, Parsons JR. Bone graft and bone graft substitutes: a review of current technology and applications. J Appl Biomater. 1991;2:187–208. - PubMed
-
- Arrington ED, Smith WJ, Chambers HG, et al. Complications of iliac crest bone graft harvesting. Clin Orthop Relat Res. 1996;329:300–309. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

