High-mobility group box 1 from reactive astrocytes enhances the accumulation of endothelial progenitor cells in damaged white matter
- PMID: 23227954
- PMCID: PMC3604050
- DOI: 10.1111/jnc.12120
High-mobility group box 1 from reactive astrocytes enhances the accumulation of endothelial progenitor cells in damaged white matter
Abstract
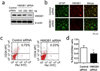
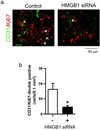
High-mobility group box 1 (HMGB1) was initially described as a damage-associated-molecular-pattern (DAMP) mediator that worsens acute brain injury after stroke. But, recent findings suggest that HMGB1 can play a surprisingly beneficial role during stroke recovery by promoting endothelial progenitor cell (EPC) function and vascular remodeling in cortical gray matter. Here, we ask whether HMGB1 may also influence EPC responses in white matter injury. The standard lysophosphatidylcholine (LPC) injection model was used to induce focal demyelination in the corpus callosum of mice. Immunostaining showed that within the focal white matter lesions, HMGB1 was up-regulated in GFAP-positive reactive astrocytes, along with the accumulation of Flk1/CD34-double-positive EPCs that expressed pro-recovery mediators such as brain-derived neurotrophic factor and basic fibroblast growth factor. Astrocyte-EPC signaling required the HMGB1 receptor RAGE as treatment with RAGE-neutralizing antibody significantly decreased EPC accumulation. Moreover, suppression of HMGB1 with siRNA in vivo significantly decreased EPC numbers in damaged white matter as well as proliferated endothelial cell numbers. Finally, in vitro cell culture systems confirmed that HMGB1 directly affected EPC function such as migration and tube formation. Taken together, our findings suggest that HMGB1 from reactive astrocytes may attract EPCs to promote recovery after white matter injury.
Keywords: endothelial progenitor cells (EPCs); high‐mobility group box 1 (HMGB1); neurovascular unit; reactive astrocytes; white matter injury; white matter remodeling.
© 2012 International Society for Neurochemistry.
Figures
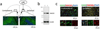


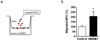
Similar articles
-
Reactive astrocytes promote adhesive interactions between brain endothelium and endothelial progenitor cells via HMGB1 and beta-2 integrin signaling.Stem Cell Res. 2014 Mar;12(2):531-8. doi: 10.1016/j.scr.2013.12.008. Epub 2013 Dec 29. Stem Cell Res. 2014. PMID: 24480450 Free PMC article.
-
Astrocytic high-mobility group box 1 promotes endothelial progenitor cell-mediated neurovascular remodeling during stroke recovery.Proc Natl Acad Sci U S A. 2012 May 8;109(19):7505-10. doi: 10.1073/pnas.1121146109. Epub 2012 Apr 23. Proc Natl Acad Sci U S A. 2012. PMID: 22529378 Free PMC article.
-
Effect of HMGB1 on the paracrine action of EPC promotes post-ischemic neovascularization in mice.Stem Cells. 2014 Oct;32(10):2679-89. doi: 10.1002/stem.1754. Stem Cells. 2014. PMID: 24888319
-
High-mobility group box 1: an amplifier of stem and progenitor cell activity after stroke.Acta Neurochir Suppl. 2013;118:31-8. doi: 10.1007/978-3-7091-1434-6_5. Acta Neurochir Suppl. 2013. PMID: 23564100 Free PMC article. Review.
-
High Mobility Group Box-1 and Blood-Brain Barrier Disruption.Cells. 2020 Dec 10;9(12):2650. doi: 10.3390/cells9122650. Cells. 2020. PMID: 33321691 Free PMC article. Review.
Cited by
-
Reactive astrocytes promote adhesive interactions between brain endothelium and endothelial progenitor cells via HMGB1 and beta-2 integrin signaling.Stem Cell Res. 2014 Mar;12(2):531-8. doi: 10.1016/j.scr.2013.12.008. Epub 2013 Dec 29. Stem Cell Res. 2014. PMID: 24480450 Free PMC article.
-
Near infrared light amplifies endothelial progenitor cell accumulation after stroke.Cond Med. 2019 Aug;2(4):170-177. Cond Med. 2019. PMID: 34291201 Free PMC article.
-
The Roles of High Mobility Group Box 1 in Cerebral Ischemic Injury.Front Cell Neurosci. 2020 Dec 15;14:600280. doi: 10.3389/fncel.2020.600280. eCollection 2020. Front Cell Neurosci. 2020. PMID: 33384585 Free PMC article. Review.
-
Unveiling the hidden culprit: How the brain-gut axis fuels neuroinflammation in ischemic stroke.Surg Neurol Int. 2024 Nov 1;15:394. doi: 10.25259/SNI_703_2024. eCollection 2024. Surg Neurol Int. 2024. PMID: 39640340 Free PMC article.
-
Stem Cell Therapy for Neuroprotection in the Growth-Restricted Newborn.Stem Cells Transl Med. 2022 Apr 29;11(4):372-382. doi: 10.1093/stcltm/szac005. Stem Cells Transl Med. 2022. PMID: 35485440 Free PMC article. Review.
References
-
- Carmichael ST. Cellular and molecular mechanisms of neural repair after stroke: making waves. Ann Neurol. 2006;59:735–742. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

