Alzheimer disease periventricular white matter lesions exhibit specific proteomic profile alterations
- PMID: 23231993
- PMCID: PMC3568229
- DOI: 10.1016/j.neuint.2012.12.001
Alzheimer disease periventricular white matter lesions exhibit specific proteomic profile alterations
Abstract
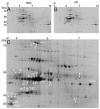
The white matter (WM) represents approximately half the cerebrum volume and is profoundly affected in Alzheimer's disease (AD). However, both the WM responses to AD as well as potential influences of this compartment to dementia pathogenesis remain comparatively neglected. Neuroimaging studies have revealed WM alterations are commonly associated with AD and renewed interest in examining the pathologic basis and importance of these changes. In AD subjects, immunohistochemistry and electron microscopy revealed changes in astrocyte morphology and myelin loss as well as up to 30% axonal loss in areas of WM rarefaction when measured against non-demented control (NDC) tissue. Comparative proteomic analyses were performed on pooled samples of periventricular WM (PVWM) obtained from AD (n=4) and NDC (n=5) subjects with both groups having a mean age of death of 86 years. All subjects had an apolipoprotein E ε3/3 genotype with the exception of one NDC subject who was ε2/3. Urea-detergent homogenates were analyzed using two different separation techniques: 2-dimensional isoelectric focusing/reverse-phase chromatography and 2-dimensional difference gel electrophoresis (2D-DIGE). Proteins with different expression levels between the 2 diagnostic groups were identified using MALDI-Tof/Tof mass spectrometry. In addition, Western blots were used to quantify proteins of interest in individual AD and NDC cases. Our proteomic studies revealed that when WM protein pools were loaded at equal amounts of total protein for comparative analyses, there were quantitative differences between the 2 groups. Molecules related to cytoskeleton maintenance, calcium metabolism and cellular survival such as glial fibrillary acidic protein, vimentin, tropomyosin, collapsin response mediator protein-2, calmodulin, S100-P, annexin A1, α-internexin, α- and β-synuclein, α-B-crystalline, fascin-1, ubiquitin carboxyl-terminal esterase and thymosine were altered between AD and NDC pools. Our experiments suggest that WM activities become globally impaired during the course of AD with significant morphological, biochemical and functional consequential implications for gray matter function and cognitive deficits. These observations may endorse the hypothesis that WM dysfunction is not only a consequence of AD pathology, but that it may precipitate and/or potentiate AD dementia.
Copyright © 2012 Elsevier Ltd. All rights reserved.
Figures


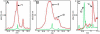

References
-
- Adams JC. Roles of fascin in cell adhesion and motility. Curr. Opin. Cell Biol. 2004;16:590–596. - PubMed
-
- Al-Hasani OH, Smith C. Traumatic white matter injury and toxic leukoencephalopathies. Expert. Rev. Neurother. 2011;11:1315–1324. - PubMed
-
- Arumugam T, Simeone DM, Schmidt AM, Logsdon CD. S100P stimulates cell proliferation and survival via receptor for activated glycation end products (RAGE) J. Biol. Chem. 2004;279:5059–5065. - PubMed
-
- Bartzokis G. Age-related myelin breakdown: a developmental model of cognitive decline and Alzheimer’s disease. Neurobiol. Aging. 2004;25:5–18. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

