βIII spectrin regulates the structural integrity and the secretory protein transport of the Golgi complex
- PMID: 23233669
- PMCID: PMC3554888
- DOI: 10.1074/jbc.M112.406462
βIII spectrin regulates the structural integrity and the secretory protein transport of the Golgi complex
Abstract
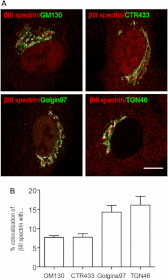
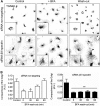
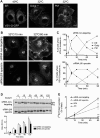
A spectrin-based cytoskeleton is associated with endomembranes, including the Golgi complex and cytoplasmic vesicles, but its role remains poorly understood. Using new generated antibodies to specific peptide sequences of the human βIII spectrin, we here show its distribution in the Golgi complex, where it is enriched in the trans-Golgi and trans-Golgi network. The use of a drug-inducible enzymatic assay that depletes the Golgi-associated pool of PI4P as well as the expression of PH domains of Golgi proteins that specifically recognize this phosphoinositide both displaced βIII spectrin from the Golgi. However, the interference with actin dynamics using actin toxins did not affect the localization of βIII spectrin to Golgi membranes. Depletion of βIII spectrin using siRNA technology and the microinjection of anti-βIII spectrin antibodies into the cytoplasm lead to the fragmentation of the Golgi. At ultrastructural level, Golgi fragments showed swollen distal Golgi cisternae and vesicular structures. Using a variety of protein transport assays, we show that the endoplasmic reticulum-to-Golgi and post-Golgi protein transports were impaired in βIII spectrin-depleted cells. However, the internalization of the Shiga toxin subunit B to the endoplasmic reticulum was unaffected. We state that βIII spectrin constitutes a major skeletal component of distal Golgi compartments, where it is necessary to maintain its structural integrity and secretory activity, and unlike actin, PI4P appears to be highly relevant for the association of βIII spectrin the Golgi complex.
Figures


Similar articles
-
A widely expressed betaIII spectrin associated with Golgi and cytoplasmic vesicles.Proc Natl Acad Sci U S A. 1998 Nov 24;95(24):14158-63. doi: 10.1073/pnas.95.24.14158. Proc Natl Acad Sci U S A. 1998. PMID: 9826670 Free PMC article.
-
beta III spectrin binds to the Arp1 subunit of dynactin.J Biol Chem. 2001 Sep 28;276(39):36598-605. doi: 10.1074/jbc.M104838200. Epub 2001 Jul 18. J Biol Chem. 2001. PMID: 11461920
-
Actin microfilaments facilitate the retrograde transport from the Golgi complex to the endoplasmic reticulum in mammalian cells.Traffic. 2001 Oct;2(10):717-26. doi: 10.1034/j.1600-0854.2001.21006.x. Traffic. 2001. PMID: 11576448
-
Speculating about spectrin: new insights into the Golgi-associated cytoskeleton.Trends Cell Biol. 1998 Jan;8(1):26-9. doi: 10.1016/s0962-8924(97)01195-1. Trends Cell Biol. 1998. PMID: 9695804 Review.
-
A spectrin membrane skeleton of the Golgi complex.Biochim Biophys Acta. 1998 Aug 14;1404(1-2):153-60. doi: 10.1016/s0167-4889(98)00054-8. Biochim Biophys Acta. 1998. PMID: 9714784 Review.
Cited by
-
Actin acting at the Golgi.Histochem Cell Biol. 2013 Sep;140(3):347-60. doi: 10.1007/s00418-013-1115-8. Epub 2013 Jun 27. Histochem Cell Biol. 2013. PMID: 23807268 Review.
-
SPTBN2 suppresses ferroptosis in NSCLC cells by facilitating SLC7A11 membrane trafficking and localization.Redox Biol. 2024 Apr;70:103039. doi: 10.1016/j.redox.2024.103039. Epub 2024 Jan 14. Redox Biol. 2024. PMID: 38241838 Free PMC article.
-
The Spread of Spectrin in Ataxia and Neurodegenerative Disease.J Exp Neurol. 2021;2(3):131-139. J Exp Neurol. 2021. PMID: 34528024 Free PMC article.
-
Emerging roles of phosphatidylinositol 4-phosphate and phosphatidylinositol 4,5-bisphosphate as regulators of multiple steps in autophagy.J Biochem. 2020 Oct 1;168(4):329-336. doi: 10.1093/jb/mvaa089. J Biochem. 2020. PMID: 32745205 Free PMC article. Review.
-
Spectrin-based membrane skeleton supports ciliogenesis.PLoS Biol. 2019 Jul 12;17(7):e3000369. doi: 10.1371/journal.pbio.3000369. eCollection 2019 Jul. PLoS Biol. 2019. PMID: 31299042 Free PMC article.
References
-
- Delaunay J. (2007) The molecular basis of hereditary red cell membrane disorders. Blood Rev. 21, 1–20 - PubMed
-
- Bennett V., Healy J. (2008) Organizing the fluid membrane bilayer: diseases linked to spectrin and ankyrin. Trends Mol. Med. 14, 28–36 - PubMed
-
- Bennett V. (1989) The spectrin-actin junction of erythrocyte membrane skeletons. Biochim. Biophys. Acta 988, 107–121 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

