Development of ex vivo organ culture models to mimic human corneal scarring
- PMID: 23233791
- PMCID: PMC3519381
Development of ex vivo organ culture models to mimic human corneal scarring
Abstract
Purpose: To develop ex vivo organ culture models of human corneal scarring suitable for pharmacological testing and the study of the molecular mechanisms leading to corneal haze after laser surgery or wounding.
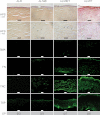
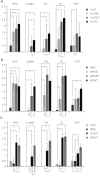
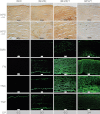
Methods: Corneas from human donors were cultured ex vivo for 30 days, either at the air-liquid interface (AL) or immersed (IM) in the culture medium. Histological features and immunofluorescence for fibronectin, tenascin C, thrombospondin-1, and α-smooth muscle actin were graded from 0 to 3 for control corneas and for corneas wounded with an excimer laser. The effects of adding 10 ng/ml transforming growth factor-β1 (TGF-β1) to the culture medium and of prior complete removal of the epithelium and limbus, thus preventing reepithelialization, were also analyzed on wounded corneas. Collagen III expression was detected with real-time PCR.
Results: Wounding alone was sufficient to induce keratocyte activation and stromal disorganization, but it was only in the presence of added TGF-β1 that intense staining for fibronectin and tenascin C was found in the AL and IM models (as well as thrombospondin-1 in the AL model) and that α-smooth muscle actin became detectable. The scar-like appearance of the corneas was exacerbated when TGF-β1 was added and reepithelialization was prevented, resulting in the majority of corneas becoming opaque and marked upregulation of collagen III.
Conclusions: THE MAIN FEATURES OF CORNEAL SCARRING WERE REPRODUCED IN THESE TWO COMPLEMENTARY MODELS: the AL model preserved differentiation of the epithelium and permits the topical application of active molecules, while the IM model ensures better perfusion by soluble compounds.
Figures


References
-
- Hayashida Y, Nishida K, Yamato M, Yang J, Sugiyama H, Watanabe K, Hori Y, Maeda N, Kikuchi A, Okano T, Tano Y. Transplantation of tissue-engineered epithelial cell sheets after excimer laser photoablation reduces postoperative corneal haze. Invest Ophthalmol Vis Sci. 2006;47:552–7. - PubMed
-
- Pellegrini G, Rama P, Mavilio F, De LM. Epithelial stem cells in corneal regeneration and epidermal gene therapy. J Pathol. 2009;217:217–28. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
