Targeting fatty acid binding protein (FABP) anandamide transporters - a novel strategy for development of anti-inflammatory and anti-nociceptive drugs
- PMID: 23236415
- PMCID: PMC3517626
- DOI: 10.1371/journal.pone.0050968
Targeting fatty acid binding protein (FABP) anandamide transporters - a novel strategy for development of anti-inflammatory and anti-nociceptive drugs
Abstract
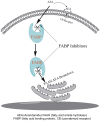
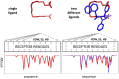
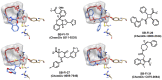
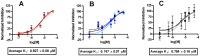
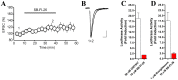
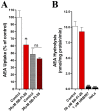
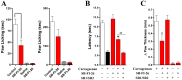
Fatty acid binding proteins (FABPs), in particular FABP5 and FABP7, have recently been identified by us as intracellular transporters for the endocannabinoid anandamide (AEA). Furthermore, animal studies by others have shown that elevated levels of endocannabinoids resulted in beneficial pharmacological effects on stress, pain and inflammation and also ameliorate the effects of drug withdrawal. Based on these observations, we hypothesized that FABP5 and FABP7 would provide excellent pharmacological targets. Thus, we performed a virtual screening of over one million compounds using DOCK and employed a novel footprint similarity scoring function to identify lead compounds with binding profiles similar to oleic acid, a natural FABP substrate. Forty-eight compounds were purchased based on their footprint similarity scores (FPS) and assayed for biological activity against purified human FABP5 employing a fluorescent displacement-binding assay. Four compounds were found to exhibit approximately 50% inhibition or greater at 10 µM, as good as or better inhibitors of FABP5 than BMS309403, a commercially available inhibitor. The most potent inhibitor, γ-truxillic acid 1-naphthyl ester (ChemDiv 8009-2334), was determined to have K(i) value of 1.19±0.01 µM. Accordingly a novel α-truxillic acid 1-naphthyl mono-ester (SB-FI-26) was synthesized and assayed for its inhibitory activity against FABP5, wherein SB-FI-26 exhibited strong binding (K(i) 0.93±0.08 µM). Additionally, we found SB-FI-26 to act as a potent anti-nociceptive agent with mild anti-inflammatory activity in mice, which strongly supports our hypothesis that the inhibition of FABPs and subsequent elevation of anandamide is a promising new approach to drug discovery. Truxillic acids and their derivatives were also shown by others to have anti-inflammatory and anti-nociceptive effects in mice and to be the active component of Chinese a herbal medicine (Incarvillea sinensis) used to treat rheumatism and pain in humans. Our results provide a likely mechanism by which these compounds exert their effects.
Conflict of interest statement
Figures

Similar articles
-
SAR studies on truxillic acid mono esters as a new class of antinociceptive agents targeting fatty acid binding proteins.Eur J Med Chem. 2018 Jun 25;154:233-252. doi: 10.1016/j.ejmech.2018.04.050. Epub 2018 May 26. Eur J Med Chem. 2018. PMID: 29803996 Free PMC article.
-
Inhibition of fatty acid binding proteins elevates brain anandamide levels and produces analgesia.PLoS One. 2014 Apr 4;9(4):e94200. doi: 10.1371/journal.pone.0094200. eCollection 2014. PLoS One. 2014. PMID: 24705380 Free PMC article.
-
The Antinociceptive Agent SBFI-26 Binds to Anandamide Transporters FABP5 and FABP7 at Two Different Sites.Biochemistry. 2017 Jul 11;56(27):3454-3462. doi: 10.1021/acs.biochem.7b00194. Epub 2017 Jun 28. Biochemistry. 2017. PMID: 28632393 Free PMC article.
-
Anandamide uptake explained?Trends Pharmacol Sci. 2012 Apr;33(4):181-5. doi: 10.1016/j.tips.2012.01.001. Epub 2012 Jan 31. Trends Pharmacol Sci. 2012. PMID: 22297258 Review.
-
Transport of endocannabinoids across the plasma membrane and within the cell.FEBS J. 2013 May;280(9):1895-904. doi: 10.1111/febs.12212. Epub 2013 Mar 18. FEBS J. 2013. PMID: 23441874 Review.
Cited by
-
A molecular evolution algorithm for ligand design in DOCK.J Comput Chem. 2022 Nov 5;43(29):1942-1963. doi: 10.1002/jcc.26993. Epub 2022 Sep 8. J Comput Chem. 2022. PMID: 36073674 Free PMC article.
-
Multiple endocannabinoid-mediated mechanisms in the regulation of energy homeostasis in brain and peripheral tissues.Cell Mol Life Sci. 2019 Apr;76(7):1341-1363. doi: 10.1007/s00018-018-2994-6. Epub 2019 Jan 1. Cell Mol Life Sci. 2019. PMID: 30599065 Free PMC article. Review.
-
Stereoselective, Ruthenium-Photocatalyzed Synthesis of 1,2-Diaminotruxinic Bis-amino Acids from 4-Arylidene-5(4H)-oxazolones.J Org Chem. 2022 Mar 4;87(5):3529-3545. doi: 10.1021/acs.joc.1c03092. Epub 2022 Feb 10. J Org Chem. 2022. PMID: 35143202 Free PMC article.
-
Identification of Fatty Acid Binding Protein 5 Inhibitors Through Similarity-Based Screening.Biochemistry. 2019 Oct 22;58(42):4304-4316. doi: 10.1021/acs.biochem.9b00625. Epub 2019 Oct 11. Biochemistry. 2019. PMID: 31539229 Free PMC article.
-
Fatty acid-binding protein 5 (FABP5)-related signal transduction pathway in castration-resistant prostate cancer cells: a potential therapeutic target.Precis Clin Med. 2019 Oct;2(3):192-196. doi: 10.1093/pcmedi/pbz015. Epub 2019 Sep 9. Precis Clin Med. 2019. PMID: 35694437 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

