Signal transduction pathways (MAPKs, NF-κB, and C/EBP) regulating COX-2 expression in nasal fibroblasts from asthma patients with aspirin intolerance
- PMID: 23240010
- PMCID: PMC3519844
- DOI: 10.1371/journal.pone.0051281
Signal transduction pathways (MAPKs, NF-κB, and C/EBP) regulating COX-2 expression in nasal fibroblasts from asthma patients with aspirin intolerance
Abstract
Background: Recent studies have revealed that cyclooxygenase-2 (COX-2) expression is down-regulated in aspirin-induced asthma (AIA). Various signal pathways (MAPKs, NF-κB and C/EBP) are involved in COX-2 regulation.
Objective: To investigate the regulation of COX-2 expression through MAP-kinase pathway activation and nuclear factor translocation in aspirin-induced asthma (AIA).
Methods: Fibroblasts were isolated from specimens of nasal mucosa (NM, N = 5) and nasal polyps (NP, N = 5). After IL-1β (1 ng/ml) incubation, COX-2 and phosphorylated forms of ERK, JNK and p38 MAPK were measured by Western blot. MAPK's role in IL-1β-induced COX-2 expression was assessed by treating cells with ERK (PD98059), JNK (SP600125) and p38 MAPK (SB203580) inhibitors (0.1-10 µM) prior to IL-1β exposure. NF-κB and C/EBP nuclear translocation was measured by Western blot and TransAM® after IL-1β (10 ng/ml) exposure.
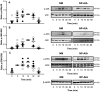
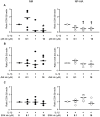
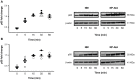
Results: No differences were observed in the MAPK phosphorylation time-course between NM and NP-AIA fibroblasts. The p38 MAPK inhibitor at 10 µM significantly reduced IL-1β-induced COX-2 expression in NM fibroblasts (85%). In NP-AIA fibroblasts the COX-2 inhibition (65%) at 1 and 10 µM was not statistically significant compared to non-treated cells. ERK and JNK inhibitors had no significant effect in either the NM or NP-AIA cultures. The effect of IL-1β on NF-κB and C/EBP subunits' nuclear translocation was similar between NM and NP-AIA fibroblasts.
Conclusions: These results suggest that p38 MAPK is the only MAPK involved in IL-1β-induced COX-2 expression. NM and NP-AIA fibroblasts have similar MAPK phosphorylation dynamics and nuclear factor translocation (NF-κB and C/EBP). COX-2 downregulation observed in AIA patients appears not to be caused by differences in MAPK dynamics or transcription factor translocation.
Conflict of interest statement
Figures

Similar articles
-
Low prostaglandin E2 and cyclooxygenase expression in nasal mucosa fibroblasts of aspirin-intolerant asthmatics.Respirology. 2013 May;18(4):711-7. doi: 10.1111/resp.12076. Respirology. 2013. PMID: 23441755
-
Low E-prostanoid 2 receptor levels and deficient induction of the IL-1β/IL-1 type I receptor/COX-2 pathway: Vicious circle in patients with aspirin-exacerbated respiratory disease.J Allergy Clin Immunol. 2016 Jan;137(1):99-107.e7. doi: 10.1016/j.jaci.2015.09.028. Epub 2015 Nov 10. J Allergy Clin Immunol. 2016. PMID: 26560040
-
Non-Transcriptional and Translational Function of Canonical NF-κB Signaling in Activating ERK1/2 in IL-1β-Induced COX-2 Expression in Synovial Fibroblasts.Front Immunol. 2020 Oct 7;11:579266. doi: 10.3389/fimmu.2020.579266. eCollection 2020. Front Immunol. 2020. PMID: 33117381 Free PMC article.
-
IL-17 stimulates MMP-1 expression in primary human cardiac fibroblasts via p38 MAPK- and ERK1/2-dependent C/EBP-beta , NF-kappaB, and AP-1 activation.Am J Physiol Heart Circ Physiol. 2007 Dec;293(6):H3356-65. doi: 10.1152/ajpheart.00928.2007. Epub 2007 Oct 5. Am J Physiol Heart Circ Physiol. 2007. PMID: 17921324
-
Anti-Inflammatory Effects of Hypophyllanthin and Niranthin Through Downregulation of NF-κB/MAPKs/PI3K-Akt Signaling Pathways.Inflammation. 2018 Jun;41(3):984-995. doi: 10.1007/s10753-018-0752-4. Inflammation. 2018. PMID: 29427163
Cited by
-
Defective Induction of COX-2 Expression by Psoriatic Fibroblasts Promotes Pro-inflammatory Activation of Macrophages.Front Immunol. 2019 Mar 20;10:536. doi: 10.3389/fimmu.2019.00536. eCollection 2019. Front Immunol. 2019. PMID: 30984165 Free PMC article.
-
Clinical efficacy of recombinant human latrophilin 3 antibody in the treatment of pediatric asthma.Exp Ther Med. 2018 Jan;15(1):539-547. doi: 10.3892/etm.2017.5376. Epub 2017 Oct 25. Exp Ther Med. 2018. PMID: 29375702 Free PMC article.
-
Release of cyclooxygenase-2 and lipoxin A4 from blood leukocytes in aspirin-exacerbated respiratory disease.Allergy Rhinol (Providence). 2016 Jan 1;7(3):158-163. doi: 10.2500/ar.2016.7.0172. Allergy Rhinol (Providence). 2016. PMID: 28107149 Free PMC article.
-
Sphingolipids and antimicrobial peptides: function and roles in atopic dermatitis.Biomol Ther (Seoul). 2013 Jul 30;21(4):251-7. doi: 10.4062/biomolther.2013.058. Biomol Ther (Seoul). 2013. PMID: 24244808 Free PMC article. Review.
References
-
- Stevenson DD, Szczeklik A (2006) Clinical and pathologic perspectives on aspirin sensitivity and asthma. J Allergy Clin Immunol 118: 773–86. - PubMed
-
- Fokkens WJ, Lund VJ, Mullol J, Bachert C, Alobid I, et al. (2012) EPOS 2012: European position paper on rhinosinusitis and nasal polyps 2012. A summary for otorhinolaryngologists. Rhinology 50: 1–12. - PubMed
-
- Picado C (2006) Mechanisms of aspirin sensitivity. Curr Allegy Asthma Rep 6: 198–202. - PubMed
-
- Kowalski ML, Pawliczak R, Wozniak J, Siuda K, Poniatowska M, et al. (2000) Differential metabolism of arachidonic acid in nasal polyp epithelial cells cultured from aspirin-sensitive and aspirin-tolerant patients. Am J Respir Crit Care Med 16: 391–8. - PubMed
-
- Schäfer D, Schmid M, Göde UC, Baenkler HW (1999) Dynamics of eicosanoids in peripheral blood cells during bronchial provocation in aspirin-intolerant asthmatics. Eur Respir J 13: 638–46. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

