The Toxoplasma nuclear factor TgAP2XI-4 controls bradyzoite gene expression and cyst formation
- PMID: 23240624
- PMCID: PMC3556193
- DOI: 10.1111/mmi.12121
The Toxoplasma nuclear factor TgAP2XI-4 controls bradyzoite gene expression and cyst formation
Abstract
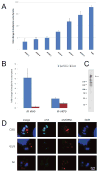
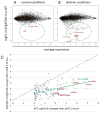
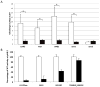
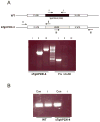
Toxoplasma gondii undergoes many phenotypic changes during its life cycle. The recent identification of AP2 transcription factors in T. gondii has provided a platform for studying the mechanisms controlling gene expression. In the present study, we report that a recombinant protein encompassing the TgAP2XI-4 AP2 domain was able to specifically bind to a DNA motif using gel retardation assays. TgAP2XI-4 protein is localized in the parasite nucleus throughout the tachyzoite life cycle in vitro, with peak expression occurring after cytokinesis. We found that the TgAP2XI-4 transcript level was higher in bradyzoite cysts isolated from brains of chronically infected mice than in the rapidly replicating tachyzoites. A knockout of the TgAP2XI-4 gene in both T. gondii virulent type I and avirulent type II strains reveals its role in modulating expression and promoter activity of genes involved in stage conversion of the rapidly replicating tachyzoites to the dormant cyst forming bradyzoites. Furthermore, mice infected with the type II KO mutants show a drastically reduced brain cyst burden. Thus, our results validate TgAP2XI-4 as a novel nuclear factor that regulates bradyzoite gene expression during parasite differentiation and cyst formation.
© 2012 Blackwell Publishing Ltd.
Figures


Similar articles
-
Toxoplasma transcription factor TgAP2XI-5 regulates the expression of genes involved in parasite virulence and host invasion.J Biol Chem. 2013 Oct 25;288(43):31127-38. doi: 10.1074/jbc.M113.486589. Epub 2013 Sep 10. J Biol Chem. 2013. PMID: 24025328 Free PMC article.
-
Toxoplasma gondii metacaspase 2 is an important factor that influences bradyzoite formation in the Pru strain.Parasitol Res. 2020 Jul;119(7):2287-2298. doi: 10.1007/s00436-020-06722-3. Epub 2020 May 29. Parasitol Res. 2020. PMID: 32468190
-
ApiAP2 transcription factor restricts development of the Toxoplasma tissue cyst.Proc Natl Acad Sci U S A. 2013 Apr 23;110(17):6871-6. doi: 10.1073/pnas.1300059110. Epub 2013 Apr 9. Proc Natl Acad Sci U S A. 2013. PMID: 23572590 Free PMC article.
-
The Epigenome, Cell Cycle, and Development in Toxoplasma.Annu Rev Microbiol. 2018 Sep 8;72:479-499. doi: 10.1146/annurev-micro-090817-062741. Epub 2018 Jun 22. Annu Rev Microbiol. 2018. PMID: 29932347 Review.
-
Toxoplasma gondii: determinants of tachyzoite to bradyzoite conversion.Parasitol Res. 2010 Jul;107(2):253-60. doi: 10.1007/s00436-010-1899-6. Epub 2010 Jun 1. Parasitol Res. 2010. PMID: 20514494 Free PMC article. Review.
Cited by
-
Transcriptional repression by ApiAP2 factors is central to chronic toxoplasmosis.PLoS Pathog. 2018 May 2;14(5):e1007035. doi: 10.1371/journal.ppat.1007035. eCollection 2018 May. PLoS Pathog. 2018. PMID: 29718996 Free PMC article.
-
Constitutive upregulation of transcription factors underlies permissive bradyzoite differentiation in a natural isolate of Toxoplasma gondii.bioRxiv [Preprint]. 2024 Feb 28:2024.02.28.582596. doi: 10.1101/2024.02.28.582596. bioRxiv. 2024. Update in: mBio. 2024 Sep 11;15(9):e0064124. doi: 10.1128/mbio.00641-24. PMID: 38464000 Free PMC article. Updated. Preprint.
-
Molecular mechanisms of surface antigen suppression by ApiAP2 and its implications for vaccine development.Vet Res. 2025 Mar 22;56(1):63. doi: 10.1186/s13567-025-01491-2. Vet Res. 2025. PMID: 40121506 Free PMC article.
-
The transcription factor AP2XI-2 is a key negative regulator of Toxoplasma gondii merogony.Nat Commun. 2024 Jan 26;15(1):793. doi: 10.1038/s41467-024-44967-z. Nat Commun. 2024. PMID: 38278808 Free PMC article.
-
MAG2, a Toxoplasma gondii Bradyzoite Stage-Specific Cyst Matrix Protein.mSphere. 2020 Feb 19;5(1):e00100-20. doi: 10.1128/mSphere.00100-20. mSphere. 2020. PMID: 32075884 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

