Platelet IκB kinase-β deficiency increases mouse arterial neointima formation via delayed glycoprotein Ibα shedding
- PMID: 23241410
- PMCID: PMC3755353
- DOI: 10.1161/ATVBAHA.112.300781
Platelet IκB kinase-β deficiency increases mouse arterial neointima formation via delayed glycoprotein Ibα shedding
Abstract
Objective: On the luminal surface of injured arteries, platelet activation and leukocyte-platelet interactions are critical for the initiation and progression of arterial restenosis. The transcription factor nuclear factor-κB is a critical molecule in platelet activation. Here, we investigated the role of the platelet nuclear factor-κB pathway in forming arterial neointima after arterial injury.
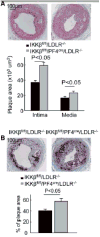
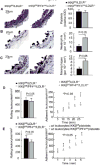
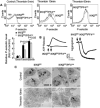
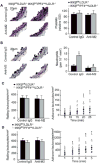
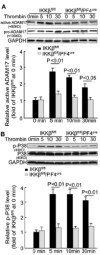
Methods and results: We performed carotid artery wire injuries in low-density lipoprotein receptor-deficient (LDLR(-/-)) mice with a platelet-specific deletion of IκB kinase-β (IKKβ) (IKKβ(fl/fl)/PF4(cre)/LDLR(-/-)) and in control mice (IKKβ(fl/fl)/LDLR(-/-)). The size of the arterial neointima was 61% larger in the IKKβ(fl/fl)/PF4(cre)/LDLR(-/-) mice compared with the littermate control IKKβ(fl/fl)/LDLR(-/-) mice. Compared with the control mice, the IKKβ(fl/fl)/PF4(cre)/LDLR(-/-) mice exhibited more leukocyte adhesion at the injured area. The extent of glycoprotein Ibα shedding after platelet activation was compromised in the IKKβ-deficient platelets. This effect was associated with a low level of the active form of A Disintegrin And Metalloproteinase 17, the key enzyme involved in mediating glycoprotein Ibα shedding in activated IKKβ-deficient platelets.
Conclusions: Platelet IKKβ deficiency increases the formation of injury-induced arterial neointima formation. Thus, nuclear factor-κB-related inhibitors should be carefully evaluated for use in patients after an arterial intervention.
Figures

Similar articles
-
Myeloid-specific IκB kinase β deficiency decreases atherosclerosis in low-density lipoprotein receptor-deficient mice.Arterioscler Thromb Vasc Biol. 2012 Dec;32(12):2869-76. doi: 10.1161/ATVBAHA.112.254573. Epub 2012 Sep 27. Arterioscler Thromb Vasc Biol. 2012. PMID: 23023371 Free PMC article.
-
Deletion of junctional adhesion molecule A from platelets increases early-stage neointima formation after wire injury in hyperlipidemic mice.J Cell Mol Med. 2017 Aug;21(8):1523-1531. doi: 10.1111/jcmm.13083. Epub 2017 Feb 17. J Cell Mol Med. 2017. PMID: 28211187 Free PMC article.
-
Deficiency of MAPK-activated protein kinase 2 (MK2) prevents adverse remodelling and promotes endothelial healing after arterial injury.Thromb Haemost. 2014 Dec;112(6):1264-76. doi: 10.1160/TH14-02-0174. Epub 2014 Aug 14. Thromb Haemost. 2014. PMID: 25120198
-
Inflammation and vascular injury: basic discovery to drug development.Circ J. 2012;76(8):1811-8. doi: 10.1253/circj.cj-12-0801. Epub 2012 Jul 30. Circ J. 2012. PMID: 22785436 Free PMC article. Review.
-
Platelet physiology and thrombosis.Thromb Res. 2004;114(5-6):447-53. doi: 10.1016/j.thromres.2004.07.020. Thromb Res. 2004. PMID: 15507277 Review.
Cited by
-
IκB kinase phosphorylation of SNAP-23 controls platelet secretion.Blood. 2013 May 30;121(22):4567-74. doi: 10.1182/blood-2012-11-470468. Epub 2013 Apr 23. Blood. 2013. PMID: 23613522 Free PMC article.
-
CD40L Priming of Platelets via NF-κB Activation is CD40- and TAK1-Dependent.J Am Heart Assoc. 2018 Dec 4;7(23):e03677. doi: 10.1161/JAHA.118.009636. J Am Heart Assoc. 2018. PMID: 30571597 Free PMC article.
-
NF-κB dynamics in the language of immune cells.Trends Immunol. 2023 Jan;44(1):32-43. doi: 10.1016/j.it.2022.11.005. Epub 2022 Dec 3. Trends Immunol. 2023. PMID: 36473794 Free PMC article. Review.
-
Large-Scale Profiling on lncRNAs in Human Platelets: Correlation with Platelet Reactivity.Cells. 2022 Jul 21;11(14):2256. doi: 10.3390/cells11142256. Cells. 2022. PMID: 35883699 Free PMC article.
-
Role of NF-κB in Platelet Function.Int J Mol Sci. 2019 Aug 27;20(17):4185. doi: 10.3390/ijms20174185. Int J Mol Sci. 2019. PMID: 31461836 Free PMC article. Review.
References
-
- Davies MG, Hagen PO. Pathobiology of intimal hyperplasia. Br J Surg. 1994;81:1254–1269. - PubMed
-
- Coller BS. Binding of abciximab to alpha v beta 3 and activated alpha m beta 2 receptors: With a review of platelet-leukocyte interactions. Thromb Haemost. 1999;82:326–336. - PubMed
-
- Schober A, Weber C. Mechanisms of monocyte recruitment in vascular repair after injury. Antioxid Redox Signal. 2005;7:1249–1257. - PubMed
-
- Manka DR, Wiegman P, Din S, Sanders JM, Green SA, Gimple LW, Ragosta M, Powers ER, Ley K, Sarembock IJ. Arterial injury increases expression of inflammatory adhesion molecules in the carotid arteries of apolipoprotein-e-deficient mice. J Vasc Res. 1999;36:372–378. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

