Effects of α-synuclein overexpression in transgenic Caenorhabditis elegans strains
- PMID: 23244416
- PMCID: PMC3744922
- DOI: 10.2174/1871527311211080005
Effects of α-synuclein overexpression in transgenic Caenorhabditis elegans strains
Abstract
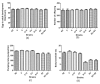
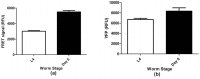
The neural protein α-synuclein aggregates both in vivo and in vitro to form insoluble fibrils that are involved in Parkinson's disease pathogenesis. We have generated α-synuclein/fluorescent-protein fusion constructs overexpressed in muscle cells of the nematode, Caenorhabdtis elegans. Green Fluorescent Protein (GFP) variants, Cerulean (C) or Venus (V), were fused to the C-terminus of human α-synuclein (S); the resultant fusion genes were designated SV and SC, plus a CV fusion as well as S, C and V singly. The aggregation behavior of the purified fusion proteins (expressed in E. coli) will be described elsewhere. These constructs were fused to a C. elegans unc-54 myosin promoter, and integrated transgenic lines generated by microinjection, λ-irradiation, and outcrossing of fluorescent progeny. All transgenic lines expressing α- synuclein showed significant reductions (p <0.05) in lifespan, motility and pharyngeal pumping, as compared to wildtype worms or lines expressing CFP and/or YFP only. We showed that CFP and YFP labels colocalised in granular inclusions throughout the body wall in transgenic lines expressing both SC and SV fusions (SC+SV), whereas SV+C worms displayed YFP-labelled inclusions on a diffuse CFP background. These findings implied that the α-synuclein moieties of these fusion proteins still aggregated together in vivo, whereas CFP or YFP moieties alone did not. This in turn suggested that Foerster Resonanace Energy Transfer (FRET) between CFP and YFP labels in α-synuclein aggregates could allow the extent of aggregation to be quantified. Accordingly, we also showed that net FRET signals increased 2- fold between L4 and adult SC+SV worms.
Figures
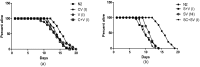

Similar articles
-
A Fluorescence Resonance Energy Transfer Assay For Monitoring α- Synclein Aggregation in a Caenorhabditis Elegans Model For Parkinson's Disease.CNS Neurol Disord Drug Targets. 2015;14(8):1054-68. doi: 10.2174/1871527314666150821110538. CNS Neurol Disord Drug Targets. 2015. PMID: 26295817
-
Microwave fields have little effect on α-synuclein aggregation in a Caenorhabditis elegans model of Parkinson's disease.Bioelectromagnetics. 2016 Feb;37(2):116-29. doi: 10.1002/bem.21959. Bioelectromagnetics. 2016. PMID: 26879225
-
Genetic Variation in Complex Traits in Transgenic α-Synuclein Strains of Caenorhabditis elegans.Genes (Basel). 2020 Jul 11;11(7):778. doi: 10.3390/genes11070778. Genes (Basel). 2020. PMID: 32664512 Free PMC article.
-
Environmental toxicants as extrinsic epigenetic factors for parkinsonism: studies employing transgenic C. elegans model.CNS Neurol Disord Drug Targets. 2012 Dec;11(8):976-83. doi: 10.2174/1871527311211080006. CNS Neurol Disord Drug Targets. 2012. PMID: 23244436 Review.
-
Intracellular pH measurements in vivo using green fluorescent protein variants.Methods Mol Biol. 2006;351:223-39. doi: 10.1385/1-59745-151-7:223. Methods Mol Biol. 2006. PMID: 16988437 Review.
Cited by
-
Small molecule inhibitors of α-synuclein oligomers identified by targeting early dopamine-mediated motor impairment in C. elegans.Mol Neurodegener. 2021 Nov 12;16(1):77. doi: 10.1186/s13024-021-00497-6. Mol Neurodegener. 2021. PMID: 34772429 Free PMC article.
-
Modeling Parkinson's Disease in C. elegans.J Parkinsons Dis. 2018;8(1):17-32. doi: 10.3233/JPD-171258. J Parkinsons Dis. 2018. PMID: 29480229 Free PMC article. Review.
-
High-Glucose Diet Attenuates the Dopaminergic Neuronal Function in C. elegans, Leading to the Acceleration of the Aging Process.ACS Omega. 2022 Sep 2;7(36):32339-32348. doi: 10.1021/acsomega.2c03384. eCollection 2022 Sep 13. ACS Omega. 2022. PMID: 36120016 Free PMC article.
-
Neuroprotective effects of a medium chain fatty acid, decanoic acid, isolated from H. leucospilota against Parkinsonism in C. elegans PD model.Front Pharmacol. 2022 Dec 13;13:1004568. doi: 10.3389/fphar.2022.1004568. eCollection 2022. Front Pharmacol. 2022. PMID: 36582526 Free PMC article.
-
2-Butoxytetrahydrofuran, Isolated from Holothuria scabra, Attenuates Aggregative and Oxidative Properties of α-Synuclein and Alleviates Its Toxicity in a Transgenic Caenorhabditis elegans Model of Parkinson's Disease.ACS Chem Neurosci. 2024 Jun 5;15(11):2182-2197. doi: 10.1021/acschemneuro.4c00008. Epub 2024 May 10. ACS Chem Neurosci. 2024. PMID: 38726817 Free PMC article.
References
-
- Dobson CM. Protein folding and misfolding. Nature. 2003;426:884–890. - PubMed
-
- Ross CA, Poirier MA. What is the role of protein aggregation in neurodegeneration? Nature Rev. Molec. Biol. 2005;6:891–898. - PubMed
-
- Eliezer D. Visualising amyloid assembly. Science. 2012;336:308–309. - PubMed
-
- Uversky V. Neuropathology, biochemistry and biophysics of α-synuclein aggregation. J. Neurochem. 2007;103:17–37. - PubMed
-
- Spillantini MG, Schmidt ML, Lee VMY, Trojanowski JQ, Jakes R, Goedert M. Alpha-synuclein in Lewy bodies. Nature. 1997;388:839– 840. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
