Development of a chiral micellar electrokinetic chromatography-tandem mass spectrometry assay for simultaneous analysis of warfarin and hydroxywarfarin metabolites: application to the analysis of patients serum samples
- PMID: 23246089
- PMCID: PMC3529819
- DOI: 10.1016/j.chroma.2012.11.046
Development of a chiral micellar electrokinetic chromatography-tandem mass spectrometry assay for simultaneous analysis of warfarin and hydroxywarfarin metabolites: application to the analysis of patients serum samples
Abstract
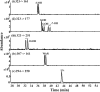
The enantioseparation of warfarin (WAR) along with the five positional and optical isomers is challenging because of the difficulty to simultaneously separate and quantitate these chiral compounds. Currently, no effective chiral CE-MS methods exist for the simultaneous enantioseparation of WAR and all its hydroxylated metabolites in a single run. Polymeric surfactants (aka. molecular micelles) are particularly compatible with micellar electrokinetic chromatography-mass spectrometry (MEKC-MS) because they have a wider elution window for enantioseparation and do not interfere with the MS detection of chiral drugs. Using polysodium N-undecenoyl-L,L-leucylvalinate (poly-L,L-SULV) as a chiral pseudophase in MEKC-MS baseline separation of WAR, its five metabolites along with the internal standard was obtained in 45 min. This is in comparison to 100 min required for separation of the same mixture with packed column CEC-MS using a vancomycin chiral stationary phase. Serum samples were extracted with mixed-mode anion-exchange (MAX) cartridge with recoveries of greater than 85.2% for all WAR and hydroxywarfarin (OH-WAR) metabolites. Utilizing the tandem MS and multiple reaction monitoring mode, the MEKC-MS/MS method was used to simultaneously generate calibration curves over a concentration range from 2 to 5000 ng/mL for R- and S-warfarin, 5 to 1000 ng/mL for R- and S-6-, 7-, 8- and 10-OH-WAR and 10 to 1000 ng/mL for R and S-4'-OH-WAR. For the first time, the limits of detection and quantitation for most WAR metabolites by MEKC-MS/MS were found to be at levels of 2 and 5 ng/mL, respectively. The method was successfully applied for the first time to analyze WAR and its metabolites in plasma samples of 55 patients undergoing WAR therapy, demonstrating the potential of chiral MEKC-MS/MS method to accurately quantitate with high sensitivity.
Copyright © 2012 Elsevier B.V. All rights reserved.
Figures
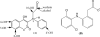







References
-
- Murray B, Coleman R, McWaters D, Ludden T, Mungall D. Ther. Drug Monit. 1987;9:1. - PubMed
-
- Rathbun S. Circulation. 2009;119:e480. - PubMed
-
- Radwan MA, Bawazeer GA, Aloudah NM, AlQuadeib BT, Aboul-Enein HY. Biomed. Chromatogr. 2012;26:6. - PubMed
-
- Hirsh J, Dalen J, Anderson DR, Poller L, Bussey H, Ansell J, Deykin D. Chest. 2001;119:8S. - PubMed
-
- Toon S, Low LK, Gibaldi M, Trager WF, O'Reilly RA, Motley CH, Goulart DA. Clin. Pharmacol. Ther. 1986;39:15. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

