Sphingolipid synthesis and scavenging in the intracellular apicomplexan parasite, Toxoplasma gondii
- PMID: 23246819
- PMCID: PMC3629565
- DOI: 10.1016/j.molbiopara.2012.11.007
Sphingolipid synthesis and scavenging in the intracellular apicomplexan parasite, Toxoplasma gondii
Abstract
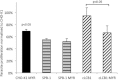
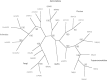
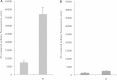
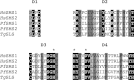
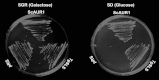
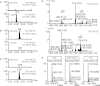
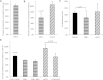
Sphingolipids are essential components of eukaryotic cell membranes, particularly the plasma membrane, and are involved in a diverse array of signal transduction pathways. Mammals produce sphingomyelin (SM) as the primary complex sphingolipid via the well characterised SM synthase. In contrast yeast, plants and some protozoa utilise an evolutionarily related inositol phosphorylceramide (IPC) synthase to synthesise IPC. This activity has no mammalian equivalent and IPC synthase has been proposed as a target for anti-fungals and anti-protozoals. However, detailed knowledge of the sphingolipid biosynthetic pathway of the apicomplexan protozoan parasites was lacking. In this study bioinformatic analyses indicated a single copy orthologue of the putative SM synthase from the apicomplexan Plasmodium falciparum (the causative agent of malaria) was a bona fide sphingolipid synthase in the related model parasite, Toxoplasma gondii (TgSLS). Subsequently, TgSLS was indicated, by complementation of a mutant cell line, to be a functional orthologue of the yeast IPC synthase (AUR1p), demonstrating resistance to the well characterised AUR1p inhibitor aureobasidin A. In vitro, recombinant TgSLS exhibited IPC synthase activity and, for the first time, the presence of IPC was demonstrated in T. gondii lipid extracts by mass spectrometry. Furthermore, host sphingolipid biosynthesis was indicated to influence, but be non-essential for, T. gondii proliferation, suggesting that whilst scavenging does take place de novo sphingolipid synthesis may be important for parasitism.
Copyright © 2012 Elsevier B.V. All rights reserved.
Figures


Similar articles
-
The Trypanosoma brucei sphingolipid synthase, an essential enzyme and drug target.Mol Biochem Parasitol. 2009 Nov;168(1):16-23. doi: 10.1016/j.molbiopara.2009.06.002. Epub 2009 Jun 21. Mol Biochem Parasitol. 2009. PMID: 19545591
-
Inhibitory effect of aureobasidin A on Toxoplasma gondii.Antimicrob Agents Chemother. 2005 May;49(5):1794-801. doi: 10.1128/AAC.49.5.1794-1801.2005. Antimicrob Agents Chemother. 2005. PMID: 15855498 Free PMC article.
-
The protozoan inositol phosphorylceramide synthase: a novel drug target that defines a new class of sphingolipid synthase.J Biol Chem. 2006 Sep 22;281(38):28200-9. doi: 10.1074/jbc.M600796200. Epub 2006 Jul 22. J Biol Chem. 2006. PMID: 16861742 Free PMC article.
-
Sphingolipids in parasitic protozoa.Adv Exp Med Biol. 2010;688:238-48. doi: 10.1007/978-1-4419-6741-1_17. Adv Exp Med Biol. 2010. PMID: 20919659 Free PMC article. Review.
-
The calcium signaling toolkit of the Apicomplexan parasites Toxoplasma gondii and Plasmodium spp.Cell Calcium. 2015 Mar;57(3):186-93. doi: 10.1016/j.ceca.2014.12.010. Epub 2014 Dec 31. Cell Calcium. 2015. PMID: 25605521 Free PMC article. Review.
Cited by
-
Ceramide biosynthesis is critical for establishment of the intracellular niche of Toxoplasma gondii.Cell Rep. 2022 Aug 16;40(7):111224. doi: 10.1016/j.celrep.2022.111224. Cell Rep. 2022. PMID: 35977499 Free PMC article.
-
Lipid metabolism: the potential targets for toxoplasmosis treatment.Parasit Vectors. 2024 Mar 6;17(1):111. doi: 10.1186/s13071-024-06213-9. Parasit Vectors. 2024. PMID: 38448975 Free PMC article. Review.
-
The antifungal Aureobasidin A and an analogue are active against the protozoan parasite Toxoplasma gondii but do not inhibit sphingolipid biosynthesis.Parasitology. 2018 Feb;145(2):148-155. doi: 10.1017/S0031182017000506. Epub 2017 May 10. Parasitology. 2018. PMID: 28486997 Free PMC article.
-
Toxoplasma ceramide synthases: Gene duplication, functional divergence, and roles in parasite fitness.FASEB J. 2023 Nov;37(11):e23229. doi: 10.1096/fj.202201603RRR. FASEB J. 2023. PMID: 37795915 Free PMC article.
-
Transcriptomic and metabolomic analyses reveal the essential nature of Rab1B in Toxoplasma gondii.Parasit Vectors. 2023 Nov 8;16(1):409. doi: 10.1186/s13071-023-06030-6. Parasit Vectors. 2023. PMID: 37941035 Free PMC article.
References
-
- Chowdhury M.N. Toxoplasmosis: a review. Journal of Medicine. 1986;17:373–396. - PubMed
-
- Dubey J.P. Toxoplasma, Hammondia, Besnotia, Sarcocystis, and other cyst-forming coccidia of man and animals. In: Kreier J.P., editor. Parasitic protozoa. Academic Press; New York: 1977. pp. 101–237.
-
- Smith W.L., Merrill A.H., Jr. Sphingolipid metabolism and signaling minireview series. Journal of Biological Chemistry. 2002;277:25841–25842. - PubMed
-
- Simons K., Ikonen E. Functional rafts in cell membranes. Nature. 1997;387:569–572. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

