Phase II study of the MEK1/MEK2 inhibitor Trametinib in patients with metastatic BRAF-mutant cutaneous melanoma previously treated with or without a BRAF inhibitor
- PMID: 23248257
- PMCID: PMC4878037
- DOI: 10.1200/JCO.2012.43.5966
Phase II study of the MEK1/MEK2 inhibitor Trametinib in patients with metastatic BRAF-mutant cutaneous melanoma previously treated with or without a BRAF inhibitor
Abstract
Purpose: BRAF mutations promote melanoma cell proliferation and survival primarily through activation of MEK. The purpose of this study was to determine the response rate (RR) for the selective, allosteric MEK1/MEK2 inhibitor trametinib (GSK1120212), in patients with metastatic BRAF-mutant melanoma.
Patients and methods: This was an open-label, two-stage, phase II study with two cohorts. Patients with metastatic BRAF-mutant melanoma previously treated with a BRAF inhibitor (cohort A) or treated with chemotherapy and/or immunotherapy (BRAF-inhibitor naive; cohort B) were enrolled. Patients received 2 mg of trametinib orally once daily.
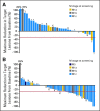
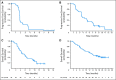
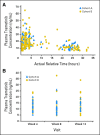
Results: In cohort A (n = 40), there were no confirmed objective responses and 11 patients (28%) with stable disease (SD); the median progression-free survival (PFS) was 1.8 months. In cohort B (n = 57), there was one (2%) complete response, 13 (23%) partial responses (PRs), and 29 patients (51%) with SD (confirmed RR, 25%); the median PFS was 4.0 months. One patient each with BRAF K601E and BRAF V600R had prolonged PR. The most frequent treatment-related adverse events for all patients were skin-related toxicity, nausea, peripheral edema, diarrhea, pruritis, and fatigue. No cutaneous squamous cell carcinoma was observed.
Conclusion: Trametinib was well tolerated. Significant clinical activity was observed in BRAF-inhibitor-naive patients previously treated with chemotherapy and/or immunotherapy. Minimal clinical activity was observed as sequential therapy in patients previously treated with a BRAF inhibitor. Together, these data suggest that BRAF-inhibitor resistance mechanisms likely confer resistance to MEK-inhibitor monotherapy. These data support further evaluation of trametinib in BRAF-inhibitor-naive BRAF-mutant melanoma, including rarer forms of BRAF-mutant melanoma.
Trial registration: ClinicalTrials.gov NCT01037127.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures
References
-
- Curtin JA, Fridlyand J, Kageshita T, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135–2147. - PubMed
-
- Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949–954. - PubMed
-
- Long GV, Menzies AM, Nagrial AM, et al. Prognostic and clinicopathologic associations of oncogenic BRAF in metastatic melanoma. J Clin Oncol. 2011;29:1239–1246. - PubMed
-
- Thomas NE, Edmiston SN, Alexander A, et al. Number of nevi and early-life ambient UV exposure are associated with BRAF-mutant melanoma. Cancer Epidemiol Biomarkers Prev. 2007;16:991–997. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

