The biochemistry of acetaminophen hepatotoxicity and rescue: a mathematical model
- PMID: 23249634
- PMCID: PMC3576299
- DOI: 10.1186/1742-4682-9-55
The biochemistry of acetaminophen hepatotoxicity and rescue: a mathematical model
Abstract
Background: Acetaminophen (N-acetyl-para-aminophenol) is the most widely used over-the-counter or prescription painkiller in the world. Acetaminophen is metabolized in the liver where a toxic byproduct is produced that can be removed by conjugation with glutathione. Acetaminophen overdoses, either accidental or intentional, are the leading cause of acute liver failure in the United States, accounting for 56,000 emergency room visits per year. The standard treatment for overdose is N-acetyl-cysteine (NAC), which is given to stimulate the production of glutathione.
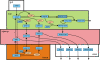
Methods: We have created a mathematical model for acetaminophen transport and metabolism including the following compartments: gut, plasma, liver, tissue, urine. In the liver compartment the metabolism of acetaminophen includes sulfation, glucoronidation, conjugation with glutathione, production of the toxic metabolite, and liver damage, taking biochemical parameters from the literature whenever possible. This model is then connected to a previously constructed model of glutathione metabolism.
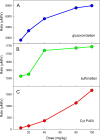
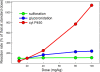
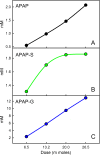
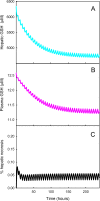
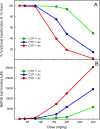
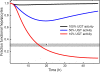
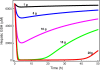
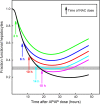
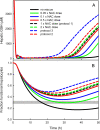
Results: We show that our model accurately reproduces published clinical and experimental data on the dose-dependent time course of acetaminophen in the plasma, the accumulation of acetaminophen and its metabolites in the urine, and the depletion of glutathione caused by conjugation with the toxic product. We use the model to study the extent of liver damage caused by overdoses or by chronic use of therapeutic doses, and the effects of polymorphisms in glucoronidation enzymes. We use the model to study the depletion of glutathione and the effect of the size and timing of N-acetyl-cysteine doses given as an antidote. Our model accurately predicts patient death or recovery depending on size of APAP overdose and time of treatment.
Conclusions: The mathematical model provides a new tool for studying the effects of various doses of acetaminophen on the liver metabolism of acetaminophen and glutathione. It can be used to study how the metabolism of acetaminophen depends on the expression level of liver enzymes. Finally, it can be used to predict patient metabolic and physiological responses to APAP doses and different NAC dosing strategies.
Figures


References
-
- Daly FF, Fountain JS, Murray L, Graudins A, Buckley NA. Guidelines for the management of paracetamol poisoning in Australia and New Zealand - explanation and elaboration. Med J Aus. 2008;188(5):296–301. - PubMed
-
- Lee WM. Acetaminophen-related acute liver failure in the United States. Heptology Res. 2008;38(supp 1):S3—S8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

