Vesicular nucleotide transporter-mediated ATP release regulates insulin secretion
- PMID: 23254199
- PMCID: PMC3548185
- DOI: 10.1210/en.2012-1818
Vesicular nucleotide transporter-mediated ATP release regulates insulin secretion
Abstract
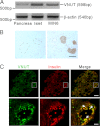
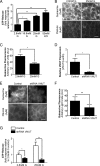
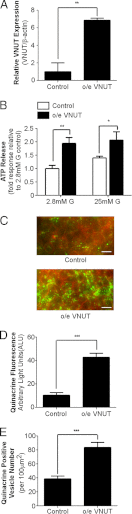
Extracellular ATP plays a critical role in regulating insulin secretion in pancreatic β cells. The ATP released from insulin secretory vesicles has been proposed to be a major source of extracellular ATP. Currently, the mechanism by which ATP accumulates into insulin secretory granules remains elusive. In this study, the authors identified the expression of a vesicular nucleotide transporter (VNUT) in mouse pancreas, isolated mouse islets, and MIN6 cells, a mouse β cell line. Immunohistochemistry and immunofluorescence revealed that VNUT colocalized extensively with insulin secretory granules. Functional studies showed that suppressing endogenous VNUT expression in β cells by small hairpin RNA knockdown greatly reduced basal- and glucose-induced ATP release. Importantly, knocking down VNUT expression by VNUT small hairpin RNA in MIN6 cells and isolated mouse islets dramatically suppressed basal insulin release and glucose-stimulated insulin secretion (GSIS). Moreover, acute pharmacologic blockade of VNUT with Evans blue, a VNUT antagonist, greatly attenuated GSIS in a dose-dependent manner. Exogenous ATP treatment effectively reversed the insulin secretion defect induced by both VNUT knockdown and functional inhibition, indicating that VNUT-mediated ATP release is essential for maintaining normal insulin secretion. In contrast to VNUT knockdown, overexpression of VNUT in β cells resulted in excessive ATP release and enhanced basal insulin secretion and GSIS. Elevated insulin secretion induced by VNUT overexpression was reversed by pharmacologic inhibition of P2X but not P2Y purinergic receptors. This study reveals VNUT is expressed in pancreatic β cells and plays an essential and novel role in regulating insulin secretion through vesicular ATP release and extracellular purinergic signaling.
Figures


References
-
- Thorens B. Molecular and cellular physiology of GLUT-2, a high-Km facilitated diffusion glucose transporter. Int Review Cytol. 1992;137:209-238 - PubMed
-
- Meglasson MD, Matschinsky FM. Pancreatic islet glucose metabolism and regulation of insulin secretion. Diabetes Metab Rev. 1986;2:163-214 - PubMed
-
- Cook DL, Hales CN. Intracellular ATP directly blocks K+ channels in pancreatic B-cells. Nature. 1984;311:271-273 - PubMed
-
- Wollheim CB, Sharp GW. Regulation of insulin release by calcium. Physiol Rev. 1981;61:914-973 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

