Function of focal adhesion kinase scaffolding to mediate endophilin A2 phosphorylation promotes epithelial-mesenchymal transition and mammary cancer stem cell activities in vivo
- PMID: 23255596
- PMCID: PMC3561552
- DOI: 10.1074/jbc.M112.420497
Function of focal adhesion kinase scaffolding to mediate endophilin A2 phosphorylation promotes epithelial-mesenchymal transition and mammary cancer stem cell activities in vivo
Abstract
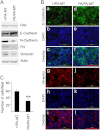
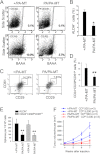
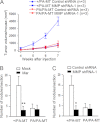
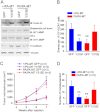
Tyrosine kinases have been shown to play critical roles in cancer development and progression, and their inhibitors hold the potential as effective targeted therapies for breast and other cancers. However, some of these kinases like focal adhesion kinase (FAK) also possess scaffolding functions in intracellular signaling, but such kinase-independent functions of FAK or other kinases have not been examined in cancer directly in vivo. Here, we report that disruption of the function of FAK scaffolding through its Pro-878/881 motif suppressed mammary tumor growth and metastasis in a well characterized murine model of human breast cancer. P878A/P881A mutation in the endogenous FAK gene decreased the expression of markers for epithelial-mesenchymal transition (EMT) and mammary cancer stem cell (MaCSC) activities in tumors derived from mutant mice. This mutation disrupted the function of FAK scaffolding to mediate endophilin A2 phosphorylation at Tyr-315 by Src, leading to the decreased surface expression of MT1-MMP, as observed previously in transformed fibroblasts in vitro. Inhibition of the downstream components of this FAK scaffolding function by Y315F endophilin A2 mutant or MT1-MMP knockdown reduced markers for EMT and MaCSC activities. Conversely, bypass of the scaffolding function using the phosphorylation mimic mutant Y315E endophilin A2 or endophilin A2 knockdown rescued the decreased markers for EMT and MaCSCs as well as surface expression of MT1-MMP in tumor cells harboring the P878A/P881A mutation. Together, these results identify a novel role of FAK scaffolding function in breast cancer, which could serve as a new target in combination with kinase inhibition for more effective treatment strategies.
Figures



Similar articles
-
Mammary epithelial-specific ablation of the focal adhesion kinase suppresses mammary tumorigenesis by affecting mammary cancer stem/progenitor cells.Cancer Res. 2009 Jan 15;69(2):466-74. doi: 10.1158/0008-5472.CAN-08-3078. Cancer Res. 2009. PMID: 19147559 Free PMC article.
-
FAK-mediated src phosphorylation of endophilin A2 inhibits endocytosis of MT1-MMP and promotes ECM degradation.Dev Cell. 2005 Aug;9(2):185-96. doi: 10.1016/j.devcel.2005.06.006. Dev Cell. 2005. PMID: 16054026
-
Distinct FAK activities determine progenitor and mammary stem cell characteristics.Cancer Res. 2013 Sep 1;73(17):5591-602. doi: 10.1158/0008-5472.CAN-13-1351. Epub 2013 Jul 5. Cancer Res. 2013. PMID: 23832665 Free PMC article.
-
Integrin signaling through FAK in the regulation of mammary stem cells and breast cancer.IUBMB Life. 2010 Apr;62(4):268-76. doi: 10.1002/iub.303. IUBMB Life. 2010. PMID: 20101634 Free PMC article. Review.
-
Signal transduction by focal adhesion kinase in cancer.Cancer Metastasis Rev. 2009 Jun;28(1-2):35-49. doi: 10.1007/s10555-008-9165-4. Cancer Metastasis Rev. 2009. PMID: 19169797 Review.
Cited by
-
Therapeutic Implication of SOCS1 Modulation in the Treatment of Autoimmunity and Cancer.Front Pharmacol. 2019 Apr 11;10:324. doi: 10.3389/fphar.2019.00324. eCollection 2019. Front Pharmacol. 2019. PMID: 31105556 Free PMC article. Review.
-
The FAK scaffold inhibitor C4 disrupts FAK-VEGFR-3 signaling and inhibits pancreatic cancer growth.Oncotarget. 2013 Oct;4(10):1632-46. doi: 10.18632/oncotarget.1365. Oncotarget. 2013. PMID: 24142503 Free PMC article.
-
A FAK scaffold inhibitor disrupts FAK and VEGFR-3 signaling and blocks melanoma growth by targeting both tumor and endothelial cells.Cell Cycle. 2014;13(16):2542-53. doi: 10.4161/15384101.2015.941760. Cell Cycle. 2014. PMID: 25486195 Free PMC article.
-
Expression of focal adhesion kinase in the eutopic endometrium of women with adenomyosis varies with dysmenorrhea and pelvic pain.Exp Ther Med. 2015 Nov;10(5):1903-1907. doi: 10.3892/etm.2015.2736. Epub 2015 Sep 9. Exp Ther Med. 2015. PMID: 26640570 Free PMC article.
-
Targeted protein degradation: A promise for undruggable proteins.Cell Chem Biol. 2021 Jul 15;28(7):934-951. doi: 10.1016/j.chembiol.2021.04.011. Epub 2021 May 17. Cell Chem Biol. 2021. PMID: 34004187 Free PMC article. Review.
References
-
- Sjöblom T., Jones S., Wood L. D., Parsons D. W., Lin J., Barber T. D., Mandelker D., Leary R. J., Ptak J., Silliman N., Szabo S., Buckhaults P., Farrell C., Meeh P., Markowitz S. D., Willis J., Dawson D., Willson J. K., Gazdar A. F., Hartigan J., Wu L., Liu C., Parmigiani G., Park B. H., Bachman K. E., Papadopoulos N., Vogelstein B., Kinzler K. W., Velculescu V. E. (2006) The consensus coding sequences of human breast and colorectal cancers. Science 314, 268–274 - PubMed
-
- Vogelstein B., Kinzler K. W. (2004) Cancer genes and the pathways they control. Nat. Med. 10, 789–799 - PubMed
-
- Parsons J. T. (2003) Focal adhesion kinase. The first ten years. J. Cell Sci. 116, 1409–1416 - PubMed
-
- Schlaepfer D. D., Mitra S. K. (2004) Multiple connections link FAK to cell motility and invasion. Curr. Opin. Genet. Dev. 14, 92–101 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

