Opposing effects of androgen deprivation and targeted therapy on prostate cancer prevention
- PMID: 23258246
- PMCID: PMC3546223
- DOI: 10.1158/2159-8290.CD-12-0262
Opposing effects of androgen deprivation and targeted therapy on prostate cancer prevention
Abstract
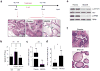
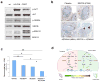
Prostate cancer is an ideal target for chemoprevention. To date, chemoprevention clinical trials with 5α-reductase inhibitors have yielded encouraging yet ultimately confounding results. Using a preclinical mouse model of high-grade prostatic intraepithelial neoplasia (HG-PIN) induced by PTEN loss, we observed unprecedented deteriorating effects of androgen deprivation, in which surgical castration or MDV3100 treatment accelerated disease progression of the otherwise stable HG-PIN to invasive castration-resistant prostate cancer (CRPC). As an alternative, targeting the phosphoinositide 3-kinase (PI3K) signaling pathway via either genetic ablation of genes encoding PI3K components or pharmacologic inhibition of the PI3K pathway reversed the PTEN loss-induced HG-PIN phenotype. Finally, concurrent inhibition of the PI3K and mitogen-activated protein kinase (MAPK) pathways was effective in blocking the growth of PTEN-null CRPC. Together, these data have revealed the potential adverse effects of antiandrogen chemoprevention in certain genetic contexts (such as PTEN loss) while showing the promise of targeted therapy in the clinical management of this complex and prevalent disease.
Significance: Chemoprevention with antiandrogen therapies is attractive for prostate cancer, given its prevalence and established hormonally mediated pathogenesis. However, because PTEN loss has been found in 9% to 45% of HG-PIN in the clinic, the current findings suggest that patients with PTEN-deficient prostate tumors might be better treated with PI3K-targeted therapies.
Conflict of interest statement
No potential conflicts of interest were disclosed by the other authors.
Figures


References
-
- Thompson IM, Goodman PJ, Tangen CM, Lucia MS, Miller GJ, Ford LG, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003;349:215–24. - PubMed
-
- Andriole GL, Bostwick DG, Brawley OW, Gomella LG, Marberger M, Montorsi F, et al. Effect of dutasteride on the risk of prostate cancer. N Engl J Med. 2010;362:1192–202. - PubMed
-
- Walsh PC. Chemoprevention of prostate cancer. N Engl J Med. 2010;362:1237–8. - PubMed
-
- Wang S, Gao J, Lei Q, Rozengurt N, Pritchard C, Jiao J, et al. Prostate-specific deletion of the murine Pten tumor suppressor gene leads to metastatic prostate cancer. Cancer Cell. 2003;4:209–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

