Hyperpolarized (13)C magnetic resonance reveals early- and late-onset changes to in vivo pyruvate metabolism in the failing heart
- PMID: 23258802
- PMCID: PMC3547367
- DOI: 10.1093/eurjhf/hfs192
Hyperpolarized (13)C magnetic resonance reveals early- and late-onset changes to in vivo pyruvate metabolism in the failing heart
Abstract
Aims: Impaired energy metabolism has been implicated in the pathogenesis of heart failure. Hyperpolarized (13)C magnetic resonance (MR), in which (13)C-labelled metabolites are followed using MR imaging (MRI) or spectroscopy (MRS), has enabled non-invasive assessment of pyruvate metabolism. We investigated the hypothesis that if we serially examined a model of heart failure using non-invasive hyperpolarized [(13)C]pyruvate with MR, the profile of in vivo pyruvate oxidation would change throughout the course of the disease.
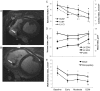
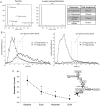
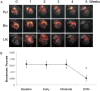
Methods and results: Dilated cardiomyopathy (DCM) was induced in pigs (n = 5) by rapid pacing. Pigs were examined using MR at weekly time points: cine-MRI assessed cardiac structure and function; hyperpolarized [2-(13)C]pyruvate was administered intravenously, and (13)C MRS monitored [(13)C]glutamate production; (31)P MRS assessed cardiac energetics [phosphocreatine (PCr)/ATP]; and hyperpolarized [1-(13)C]pyruvate was administered for MRI of pyruvate dehydrogenase complex (PDC)-mediated pyruvate oxidation via [(13)C]bicarbonate production. Early in pacing, the cardiac index decreased by 25%, PCr/ATP decreased by 26%, and [(13)C]glutamate production decreased by 51%. After clinical features of DCM appeared, end-diastolic volume increased by 40% and [(13)C]bicarbonate production decreased by 67%. Pyruvate dehydrogenase kinase 4 protein increased by two-fold, and phosphorylated Akt decreased by half. Peroxisome proliferator-activated receptor-α and carnitine palmitoyltransferase-1 gene expression decreased by a half and a third, respectively.
Conclusion: Despite early changes associated with cardiac energetics and (13)C incorporation into the Krebs cycle, pyruvate oxidation was maintained until DCM developed, when the heart's capacity to oxidize both pyruvate and fats was reduced. Hyperpolarized (13)C MR may be important to characterize metabolic changes that occur during heart failure progression.
Figures



Comment in
-
Imaging the cardiac diet.Eur J Heart Fail. 2013 Feb;15(2):123-4. doi: 10.1093/eurjhf/hfs211. Eur J Heart Fail. 2013. PMID: 23329701 No abstract available.
References
-
- Neubauer S. The failing heart—an engine out of fuel. N Engl J Med. 2007;356:1140–1151. - PubMed
-
- Stanley WC, Recchia FA, Lopaschuk GD. Myocardial substrate metabolism in the normal and failing heart. Physiol Rev. 2005;85:1093–1129. - PubMed
-
- Neubauer S, Krahe T, Schindler R, Horn M, Hillenbrand H, Entzeroth C, Mader H, Kromer EP, Riegger GA, Lackner K, Ertl G. 31P magnetic resonance spectroscopy in dilated cardiomyopathy and coronary artery disease. Altered cardiac high-energy phosphate metabolism in heart failure. Circulation. 1992;86:1810–1818. - PubMed
-
- Shen WQ, Asai K, Uechi M, Mathier MA, Shannon RP, Vatner SF, Ingwall JS. Progressive loss of myocardial ATP due to a loss of total purines during the development of heart failure in dogs—a compensatory role for the parallel loss of creatine. Circulation. 1999;100:2113–2118. - PubMed
-
- Beanlands RS, Nahmias C, Gordon E, Coates G, deKemp R, Firnau G, Fallen E. The effects of beta(1)-blockade on oxidative metabolism and the metabolic cost of ventricular work in patients with left ventricular dysfunction: a double-blind, placebo-controlled, positron-emission tomography study. Circulation. 2000;102:2070–2075. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

