NF-κB is activated in response to temozolomide in an AKT-dependent manner and confers protection against the growth suppressive effect of the drug
- PMID: 23259744
- PMCID: PMC3551789
- DOI: 10.1186/1479-5876-10-252
NF-κB is activated in response to temozolomide in an AKT-dependent manner and confers protection against the growth suppressive effect of the drug
Abstract
Background: Most DNA-damaging chemotherapeutic agents activate the transcription factor nuclear factor κB (NF-κB). However, NF-κB activation can either protect from or contribute to the growth suppressive effects of the agent. We previously showed that the DNA-methylating drug temozolomide (TMZ) activates AKT, a positive modulator of NF-κB, in a mismatch repair (MMR) system-dependent manner. Here we investigated whether NF-κB is activated by TMZ and whether AKT is involved in this molecular event. We also evaluated the functional consequence of inhibiting NF-κB on tumor cell response to TMZ.
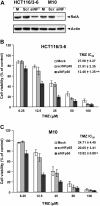
Methods: AKT phosphorylation, NF-κB transcriptional activity, IκB-α degradation, NF-κB2/p52 generation, and RelA and NF-κB2/p52 nuclear translocation were investigated in TMZ-treated MMR-deficient (HCT116, 293TLα-) and/or MMR-proficient (HCT116/3-6, 293TLα+, M10) cells. AKT involvement in TMZ-induced activation of NF-κB was addressed in HCT116/3-6 and M10 cells transiently transfected with AKT1-targeting siRNA or using the isogenic MMR-proficient cell lines pUSE2 and KD12, expressing wild type or kinase-dead mutant AKT1. The effects of inhibiting NF-κB on sensitivity to TMZ were investigated in HCT116/3-6 and M10 cells using the NF-κB inhibitor NEMO-binding domain (NBD) peptide or an anti-RelA siRNA.
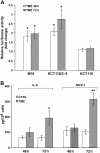
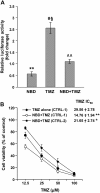
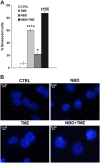
Results: TMZ enhanced NF-κB transcriptional activity, activated AKT, induced IκB-α degradation and RelA nuclear translocation in HCT116/3-6 and M10 but not in HCT116 cells. In M10 cells, TMZ promoted NF-κB2/p52 generation and nuclear translocation and enhanced the secretion of IL-8 and MCP-1. TMZ induced RelA nuclear translocation also in 293TLα+ but not in 293TLα- cells. AKT1 silencing inhibited TMZ-induced IκB-α degradation and NF-κB2/p52 generation. Up-regulation of NF-κB transcriptional activity and nuclear translocation of RelA and NF-κB2/p52 in response to TMZ were impaired in KD12 cells. RelA silencing in HCT116/3-6 and M10 cells increased TMZ-induced growth suppression. In M10 cells NBD peptide reduced basal NF-κB activity, abrogated TMZ-induced up-regulation of NF-κB activity and increased sensitivity to TMZ. In HCT116/3-6 cells, the combined treatment with NBD peptide and TMZ produced additive growth inhibitory effects.
Conclusion: NF-κB is activated in response to TMZ in a MMR- and AKT-dependent manner and confers protection against drug-induced cell growth inhibition. Our findings suggest that a clinical benefit could be obtained by combining TMZ with NF-κB inhibitors.
Figures



References
-
- Perkins ND. The diverse and complex roles of NF-kB subunits in cancer. Nat Rev Cancer. 2012;12:121–132. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

