Rationalization of allosteric pathway in Thermus sp. GH5 methylglyoxal synthase
- PMID: 23261063
- PMCID: PMC4133812
- DOI: 10.5483/bmbrep.2012.45.12.11-138
Rationalization of allosteric pathway in Thermus sp. GH5 methylglyoxal synthase
Abstract
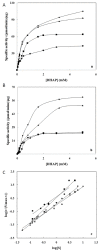
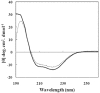
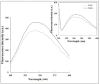
A sequence of 10 amino acids at the C-terminus region of methylglyoxal synthase from Escherichia coli (EMGS) provides an arginine, which plays a crucial role in forming a salt bridge with a proximal aspartate residue in the neighboring subunit, consequently transferring the allosteric signal between subunits. In order to verify the role of arginine, the gene encoding MGS from a thermophile species, Thermus sp. GH5 (TMGS) lacking this arginine was cloned with an additional 30 bp sequence at the 3´-end and then expressed in form of a fusion TMGS with a 10 residual segment at the C-terminus (TMGS(+)). The resulting recombinant enzyme showed a significant increase in cooperativity towards phosphate, reflected by a change in the Hill coefficient (nH) from 1.5 to 1.99. Experiments including site directed mutagenesis for Asp-10 in TMGS and TMGS(+), two dimentional structural survey, fluorescence and irreversible thermoinactivation were carried out to confirm this pathway.
Figures