Plasmonic photothermal therapy increases the tumor mass penetration of HPMA copolymers
- PMID: 23262203
- PMCID: PMC3715141
- DOI: 10.1016/j.jconrel.2012.12.007
Plasmonic photothermal therapy increases the tumor mass penetration of HPMA copolymers
Abstract
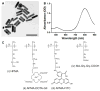
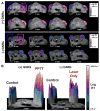
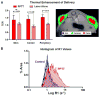
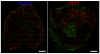
Effective drug delivery to tumors requires both transport through the vasculature and tumor interstitium. Previously, it was shown that gold nanorod (GNR) mediated plasmonic photothermal therapy (PPTT) is capable of increasing the overall accumulation of N-(2-hydroxypropyl)methacrylamide (HPMA) copolymers in prostate tumors. In the present study, it is demonstrated that PPTT is also capable of increasing the distribution of these conjugates in tumors. Gadolinium labeled HPMA copolymers were administered to mice bearing prostate tumors immediately before treatment of the right tumor with PPTT. The left tumor served as internal, untreated control. Magnetic resonance imaging (MRI) of both tumors showed that PPTT was capable of improving the tumor mass penetration of HPMA copolymers. Thermal enhancement of delivery, roughly 1.5-fold, to both the tumor center and periphery was observed. Confocal microscopy of fluorescently labeled copolymers corroborates these findings in that PPTT is capable of delivering more HPMA copolymers to the tumor's center and periphery. These results further demonstrate that PPTT is a useful tool to improve the delivery of polymer-drug conjugates.
Copyright © 2012 Elsevier B.V. All rights reserved.
Figures
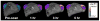

Similar articles
-
Effects of heating temperature and duration by gold nanorod mediated plasmonic photothermal therapy on copolymer accumulation in tumor tissue.Mol Pharm. 2015 May 4;12(5):1605-14. doi: 10.1021/acs.molpharmaceut.5b00019. Epub 2015 Apr 15. Mol Pharm. 2015. PMID: 25839226
-
Synergistic enhancement of cancer therapy using a combination of heat shock protein targeted HPMA copolymer-drug conjugates and gold nanorod induced hyperthermia.J Control Release. 2013 Aug 28;170(1):41-50. doi: 10.1016/j.jconrel.2013.04.006. Epub 2013 Apr 17. J Control Release. 2013. PMID: 23602864 Free PMC article.
-
Effect of radiotherapy and hyperthermia on the tumor accumulation of HPMA copolymer-based drug delivery systems.J Control Release. 2007 Feb 26;117(3):333-41. doi: 10.1016/j.jconrel.2006.10.032. Epub 2006 Nov 25. J Control Release. 2007. PMID: 17215057
-
HPMA copolymer-cyclic RGD conjugates for tumor targeting.Adv Drug Deliv Rev. 2010 Feb 17;62(2):167-83. doi: 10.1016/j.addr.2009.11.027. Epub 2009 Dec 4. Adv Drug Deliv Rev. 2010. PMID: 19951733 Review.
-
Molecular imaging of HPMA copolymers: visualizing drug delivery in cell, mouse and man.Adv Drug Deliv Rev. 2010 Feb 17;62(2):246-57. doi: 10.1016/j.addr.2009.12.007. Epub 2010 Jan 7. Adv Drug Deliv Rev. 2010. PMID: 20060431 Review.
Cited by
-
Gold nanoparticle-mediated photothermal therapy: applications and opportunities for multimodal cancer treatment.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017 Jul;9(4):10.1002/wnan.1449. doi: 10.1002/wnan.1449. Epub 2017 Feb 3. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017. PMID: 28160445 Free PMC article. Review.
-
Iron Oxide@Mesoporous Silica Core-Shell Nanoparticles as Multimodal Platforms for Magnetic Resonance Imaging, Magnetic Hyperthermia, Near-Infrared Light Photothermia, and Drug Delivery.Nanomaterials (Basel). 2023 Apr 12;13(8):1342. doi: 10.3390/nano13081342. Nanomaterials (Basel). 2023. PMID: 37110927 Free PMC article. Review.
-
Nanoscale materials for hyperthermal theranostics.Nanoscale. 2015 Apr 28;7(16):7115-26. doi: 10.1039/c4nr06164k. Nanoscale. 2015. PMID: 25816102 Free PMC article. Review.
-
Enhanced efficacy of combination heat shock targeted polymer therapeutics with high intensity focused ultrasound.Nanomedicine. 2017 Apr;13(3):1235-1243. doi: 10.1016/j.nano.2016.11.014. Epub 2016 Nov 29. Nanomedicine. 2017. PMID: 27913213 Free PMC article.
-
Recent Advances in Hydrogel-Based Phototherapy for Tumor Treatment.Gels. 2023 Apr 1;9(4):286. doi: 10.3390/gels9040286. Gels. 2023. PMID: 37102898 Free PMC article. Review.
References
-
- Duncan R. Polymer conjugates as anticancer nanomedicines. Nat Rev Cancer. 2006;6:688–701. - PubMed
-
- Duncan R, Gaspar R. Nanomedicine (s) under the microscope. Mol Pharm. 2011;8:2101–2141. - PubMed
-
- Maeda H, Wu J, Sawa T, Matsumura Y, Hori K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release. 2000;65:271–284. - PubMed
-
- Peer D, Karp JM, Hong S, Farokhzad OC, Margalit R, Langer R. Nanocarriers as an emerging platform for cancer therapy. Nat Nanotechnol. 2007;2:751–760. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

