In vivo therapeutic efficacy of chloroquine alone or in combination with primaquine against vivax malaria in Kolkata, West Bengal, India, and polymorphism in pvmdr1 and pvcrt-o genes
- PMID: 23262997
- PMCID: PMC3591881
- DOI: 10.1128/AAC.02050-12
In vivo therapeutic efficacy of chloroquine alone or in combination with primaquine against vivax malaria in Kolkata, West Bengal, India, and polymorphism in pvmdr1 and pvcrt-o genes
Abstract
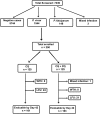
Plasmodium vivax malaria, though benign, has now become a matter of concern due to recent reports of life-threatening severity and development of parasite resistance to different antimalarial drugs. The magnitude of the problem is still undetermined. The present study was undertaken to determine the in vivo efficacy of chloroquine (CQ) and chloroquine plus primaquine in P. vivax malaria in Kolkata and polymorphisms in the pvmdr1 and pvcrt-o genes. A total of 250 patients with P. vivax monoinfection were recruited and randomized into two groups, A and B; treated with chloroquine and chloroquine plus primaquine, respectively; and followed up for 42 days according to the WHO protocol of 2009. Data were analyzed using per-protocol analyses. We assessed polymorphisms of the pvmdr1 and pvcrt-o genes by a DNA-sequencing method. Out of the 250 patients recruited, 204 completed a 42-day follow-up period, 101 in group A and 103 in group B. In group A, the non-PCR-corrected efficacy of CQ was 99% (95% confidence interval [CI], 0.944 to 1.00), and in group B, all cases were classified as adequate clinical and parasitological response (ACPR). Day 3 positivity was observed in 11 (5.3%) cases. No specific mutation pattern was recorded in the pvcrt-o gene. Eight nonsynonymous mutations were found in the pvmdr1 gene, three of which were new. The Y976F mutation was not detected in any isolate. Chloroquine, either alone or in combination with primaquine, is still effective against P. vivax malaria in the study area. (The study protocol was registered in CTRI [Clinical Trial Registry-India] of the Indian council of Medical Research under registration no. CTRI/2011/09/002031.).
Figures
References
-
- Mendis K, Sina BJ, Marchesini P, Carter R. 2001. The neglected burden of Plasmodium vivax malaria. Am. J. Trop. Med. Hyg. 64:97–106 - PubMed
-
- UK Aid, Department for International Development 31 December 2010. Malaria: burden and interventions: evidence overview. A working paper, version 1.0. Department for International Development, London, United Kingdom: http://www.dfid.gov.uk/Documents/prd/malaria-evidence-paper.pdf
-
- Guerra CA, Howes RE, Patil AP, Gething PW, Van Boeckel TP, Temperley WH, Kabaria CW, Tatem AJ, Manh BH, Elyazar IRF, Baird JK, Snow RW, Hay SI. 2010. The international limits and population at risk of Plasmodium vivax transmission in 2009. PLoS Negl. Trop. Dis. 4:e774 doi:10.1371/journal.pntd.0000774 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources