Comparison of MS/MS methods for characterization of DNA/cisplatin adducts
- PMID: 23264150
- PMCID: PMC3570664
- DOI: 10.1007/s13361-012-0532-6
Comparison of MS/MS methods for characterization of DNA/cisplatin adducts
Abstract
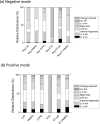
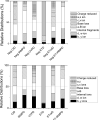
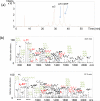
The development of activation/dissociation techniques such as ultraviolet photodissociation (UVPD), infrared multiphoton dissociation (IRMPD), and electron transfer dissociation (ETD) as alternatives to collision induced dissociation (CID) has extended the range of strategies for characterizing biologically relevant molecules. Here, we describe a comprehensive comparison of CID, IRMPD, UVPD, ETD, and hybrid processes termed ETcaD and ET-IRMPD (and analogous hybrid methods in the negative mode NETcaD and NET-IRMPD) for generating sequence-specific fragment ions and allowing adduction sites to be pinpointed for DNA/cisplatin adducts. Among the six MS/MS methods, the numerous products generated by the IRMPD and UVPD techniques resulted in the most specific and extensive backbone cleavages. We conclude that IRMPD and UVPD methods generally offer the best characteristics for pinpointing the cisplatin adduction sites in the fragment-rich spectra.
Figures


Similar articles
-
Hybrid activation methods for elucidating nucleic acid modifications.Anal Chem. 2011 Jan 1;83(1):303-10. doi: 10.1021/ac102411a. Epub 2010 Dec 9. Anal Chem. 2011. PMID: 21141922 Free PMC article.
-
Characterization of Phosphorylated Peptides by Electron-Activated and Ultraviolet Dissociation Mass Spectrometry: A Comparative Study with Collision-Induced Dissociation.J Am Soc Mass Spectrom. 2024 May 1;35(5):1040-1054. doi: 10.1021/jasms.4c00048. Epub 2024 Apr 16. J Am Soc Mass Spectrom. 2024. PMID: 38626331 Free PMC article.
-
Extensive fragmentation of pheophytin-a by infrared multiphoton dissociation tandem mass spectrometry.Rapid Commun Mass Spectrom. 2015 Dec 30;29(24):2411-8. doi: 10.1002/rcm.7391. Rapid Commun Mass Spectrom. 2015. PMID: 26563711
-
Activation of large ions in FT-ICR mass spectrometry.Mass Spectrom Rev. 2005 Mar-Apr;24(2):135-67. doi: 10.1002/mas.20012. Mass Spectrom Rev. 2005. PMID: 15389858 Review.
-
Radical-driven peptide backbone dissociation tandem mass spectrometry.Mass Spectrom Rev. 2015 Mar-Apr;34(2):116-32. doi: 10.1002/mas.21426. Epub 2014 May 26. Mass Spectrom Rev. 2015. PMID: 24863492 Review.
Cited by
-
Top-down mass spectrometry reveals multiple interactions of an acetylsalicylic acid bearing Zeise's salt derivative with peptides.J Biol Inorg Chem. 2020 Mar;25(2):285-293. doi: 10.1007/s00775-020-01760-9. Epub 2020 Feb 14. J Biol Inorg Chem. 2020. PMID: 32060649 Free PMC article.
-
Progress in Tandem Mass Spectrometry Data Analysis for Nucleic Acids.Mass Spectrom Rev. 2025 Jan 10:10.1002/mas.21923. doi: 10.1002/mas.21923. Online ahead of print. Mass Spectrom Rev. 2025. PMID: 39797409 Review.
-
Ultraviolet Photodissociation Mass Spectrometry for Analysis of Biological Molecules.Chem Rev. 2020 Apr 8;120(7):3328-3380. doi: 10.1021/acs.chemrev.9b00440. Epub 2019 Dec 18. Chem Rev. 2020. PMID: 31851501 Free PMC article. Review.
-
Recent developments in the characterization of nucleic acids by liquid chromatography, capillary electrophoresis, ion mobility, and mass spectrometry (2010-2020).J Sep Sci. 2021 Jan;44(1):340-372. doi: 10.1002/jssc.202000833. Epub 2020 Oct 15. J Sep Sci. 2021. PMID: 32974962 Free PMC article. Review.
-
Spacer Fidelity Assessments of Guide RNA by Top-Down Mass Spectrometry.ACS Cent Sci. 2023 Jul 11;9(7):1437-1452. doi: 10.1021/acscentsci.3c00289. eCollection 2023 Jul 26. ACS Cent Sci. 2023. PMID: 37521788 Free PMC article.
References
-
- Hurley LH. DNA and its associated processes as targets for cancer therapy. Nat. Rev. Cancer. 2002;2(3):188. - PubMed
-
- Ou TM, Lu YJ, Tan JH, Huang ZS, Wong KY, Gu LQ. G-quadruplexes: Targets in anticancer drug design. ChemMedChem. 2008;3(5):690. - PubMed
-
- Miller KM, Rodriguez R. G-quadruplexes: selective DNA targeting for cancer therapeutics? Expert Rev. Clin. Pharmacol. 2011;4(2):139. - PubMed
-
- Rajski SR, Williams RM. DNA cross-linking agents as antitumor drugs. Chem. Rev. 1998;98(8):2723. - PubMed
-
- Brunton LL, Lazo JS, Parker KL. Goodman & Gilman's the pharmacological basis of therapeutics. McGraw-Hill; New York: 2006.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

