Differential analysis of high-throughput quantitative genetic interaction data
- PMID: 23268787
- PMCID: PMC4056373
- DOI: 10.1186/gb-2012-13-12-r123
Differential analysis of high-throughput quantitative genetic interaction data
Abstract
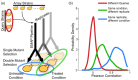
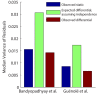
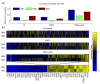
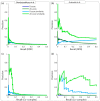
Synthetic genetic arrays have been very effective at measuring genetic interactions in yeast in a high-throughput manner and recently have been expanded to measure quantitative changes in interaction, termed 'differential interactions', across multiple conditions. Here, we present a strategy that leverages statistical information from the experimental design to produce a novel, quantitative differential interaction score, which performs favorably compared to previous differential scores. We also discuss the added utility of differential genetic-similarity in differential network analysis. Our approach is preferred for differential network analysis, and our implementation, written in MATLAB, can be found at http://chianti.ucsd.edu/~gbean/compute_differential_scores.m.
Figures

References
-
- Costanzo M, Baryshnikova A, Bellay J, Kim Y, Spear ED, Sevier CS, Ding H, Koh JLY, Toufighi K, Mostafavi S, Prinz J, St Onge RP, VanderSluis B, Makhnevych T, Vizeacoumar FJ, Alizadeh S, Bahr S, Brost RL, Chen Y, Cokol M, Deshpande R, Li Z, Lin Z-Y, Liang W, Marback M, Paw J, San Luis B-J, Shuteriqi E, Tong AHY, van Dyk N. et al.The genetic landscape of a cell. Science. 2010;13:425–431. doi: 10.1126/science.1180823. - DOI - PMC - PubMed
-
- Fiedler D, Braberg H, Mehta M, Chechik G, Cagney G, Mukherjee P, Silva AC, Shales M, Collins SR, van Wageningen S, Kemmeren P, Holstege FCP, Weissman JS, Keogh M-C, Koller D, Shokat KM, Krogan NJ. Functional organization of the S. cerevisiae phosphorylation network. Cell. 2009;13:952–963. doi: 10.1016/j.cell.2008.12.039. - DOI - PMC - PubMed
-
- Schuldiner M, Collins SR, Thompson NJ, Denic V, Bhamidipati A, Punna T, Ihmels J, Andrews B, Boone C, Greenblatt JF, Weissman JS, Krogan NJ. Exploration of the function and organization of the yeast early secretory pathway through an epistatic miniarray profile. Cell. 2005;13:507–519. doi: 10.1016/j.cell.2005.08.031. - DOI - PubMed
-
- Collins SR, Miller KM, Maas NL, Roguev A, Fillingham J, Chu CS, Schuldiner M, Gebbia M, Recht J, Shales M, Ding H, Xu H, Han J, Ingvarsdottir K, Cheng B, Andrews B, Boone C, Berger SL, Hieter P, Zhang Z, Brown GW, Ingles CJ, Emili A, Allis CD, Toczyski DP, Weissman JS, Greenblatt JF, Krogan NJ. Functional dissection of protein complexes involved in yeast chromosome biology using a genetic interaction map. Nature. 2007;13:806–810. doi: 10.1038/nature05649. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

