Missense mutations linked to friedreich ataxia have different but synergistic effects on mitochondrial frataxin isoforms
- PMID: 23269675
- PMCID: PMC3567662
- DOI: 10.1074/jbc.M112.435263
Missense mutations linked to friedreich ataxia have different but synergistic effects on mitochondrial frataxin isoforms
Abstract
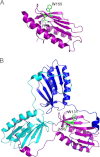
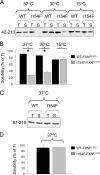
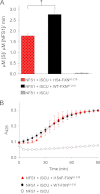
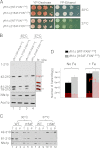
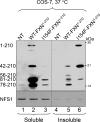
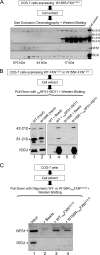
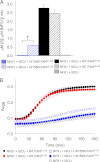
Friedreich ataxia is an early-onset multisystemic disease linked to a variety of molecular defects in the nuclear gene FRDA. This gene normally encodes the iron-binding protein frataxin (FXN), which is critical for mitochondrial iron metabolism, global cellular iron homeostasis, and antioxidant protection. In most Friedreich ataxia patients, a large GAA-repeat expansion is present within the first intron of both FRDA alleles, that results in transcriptional silencing ultimately leading to insufficient levels of FXN protein in the mitochondrial matrix and probably other cellular compartments. The lack of FXN in turn impairs incorporation of iron into iron-sulfur cluster and heme cofactors, causing widespread enzymatic deficits and oxidative damage catalyzed by excess labile iron. In a minority of patients, a typical GAA expansion is present in only one FRDA allele, whereas a missense mutation is found in the other allele. Although it is known that the disease course for these patients can be as severe as for patients with two expanded FRDA alleles, the underlying pathophysiological mechanisms are not understood. Human cells normally contain two major mitochondrial isoforms of FXN (FXN(42-210) and FXN(81-210)) that have different biochemical properties and functional roles. Using cell-free systems and different cellular models, we show that two of the most clinically severe FXN point mutations, I154F and W155R, have unique direct and indirect effects on the stability, biogenesis, or catalytic activity of FXN(42-210) and FXN(81-210) under physiological conditions. Our data indicate that frataxin point mutations have complex biochemical effects that synergistically contribute to the pathophysiology of Friedreich ataxia.
Figures

Similar articles
-
Normal and Friedreich ataxia cells express different isoforms of frataxin with complementary roles in iron-sulfur cluster assembly.J Biol Chem. 2010 Dec 3;285(49):38486-501. doi: 10.1074/jbc.M110.145144. Epub 2010 Oct 2. J Biol Chem. 2010. PMID: 20889968 Free PMC article.
-
Mitochondrial damage and senescence phenotype of cells derived from a novel frataxin G127V point mutation mouse model of Friedreich's ataxia.Dis Model Mech. 2020 Jul 27;13(7):dmm045229. doi: 10.1242/dmm.045229. Dis Model Mech. 2020. PMID: 32586831 Free PMC article.
-
Mice harboring the FXN I151F pathological point mutation present decreased frataxin levels, a Friedreich ataxia-like phenotype, and mitochondrial alterations.Cell Mol Life Sci. 2022 Jan 17;79(2):74. doi: 10.1007/s00018-021-04100-5. Cell Mol Life Sci. 2022. PMID: 35038030 Free PMC article.
-
Beyond loss of frataxin: the complex molecular pathology of Friedreich ataxia.Discov Med. 2014 Jan;17(91):25-35. Discov Med. 2014. PMID: 24411698 Review.
-
Friedreich Ataxia: From the Eye of a Molecular Biologist.Neurologist. 2015 Sep;20(3):51-5. doi: 10.1097/NRL.0000000000000054. Neurologist. 2015. PMID: 26375377 Review.
Cited by
-
Stable isotopes and LC-MS for monitoring metabolic disturbances in Friedreich's ataxia platelets.Bioanalysis. 2015;7(15):1843-55. doi: 10.4155/bio.15.118. Bioanalysis. 2015. PMID: 26295986 Free PMC article.
-
Novel Point Mutations in Frataxin Gene in Iranian Patients with Friedreich's Ataxia.Iran J Child Neurol. 2014 Winter;8(1):32-6. Iran J Child Neurol. 2014. PMID: 24665325 Free PMC article.
-
Structure of the human frataxin-bound iron-sulfur cluster assembly complex provides insight into its activation mechanism.Nat Commun. 2019 May 17;10(1):2210. doi: 10.1038/s41467-019-09989-y. Nat Commun. 2019. PMID: 31101807 Free PMC article.
-
Molecular Defects in Friedreich's Ataxia: Convergence of Oxidative Stress and Cytoskeletal Abnormalities.Front Mol Biosci. 2020 Nov 9;7:569293. doi: 10.3389/fmolb.2020.569293. eCollection 2020. Front Mol Biosci. 2020. PMID: 33263002 Free PMC article. Review.
-
Evolution of an Iron-Detoxifying Protein: Eukaryotic and Rickettsia Frataxins Contain a Conserved Site Which Is Not Present in Their Bacterial Homologues.Int J Mol Sci. 2022 Oct 29;23(21):13151. doi: 10.3390/ijms232113151. Int J Mol Sci. 2022. PMID: 36361939 Free PMC article.
References
-
- Pandolfo M. (2009) Friedreich ataxia. The clinical picture. J. Neurol. 256, 3–8 - PubMed
-
- Campuzano V., Montermini L., Lutz Y., Cova L., Hindelang C., Jiralerspong S., Trottier Y., Kish S. J., Faucheux B., Trouillas P., Authier F. J., Dürr A., Mandel J. L., Vescovi A., Pandolfo M., Koenig M. (1997) Frataxin is reduced in Friedreich ataxia patients and is associated with mitochondrial membranes. Hum. Mol. Genet. 6, 1771–1780 - PubMed
-
- Babcock M., de Silva D., Oaks R., Davis-Kaplan S., Jiralerspong S., Montermini L., Pandolfo M., Kaplan J. (1997) Regulation of mitochondrial iron accumulation by Yfh1p, a putative homolog of frataxin. Science 276, 1709–1712 - PubMed
-
- Cavadini P., Adamec J., Taroni F., Gakh O., Isaya G. (2000) Two-step processing of human frataxin by mitochondrial processing peptidase. Precursor and intermediate forms are cleaved at different rates. J. Biol. Chem. 275, 41469–41475 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

