VS-5584, a novel and highly selective PI3K/mTOR kinase inhibitor for the treatment of cancer
- PMID: 23270925
- PMCID: PMC3588144
- DOI: 10.1158/1535-7163.MCT-12-0466
VS-5584, a novel and highly selective PI3K/mTOR kinase inhibitor for the treatment of cancer
Abstract
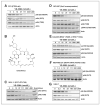
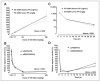
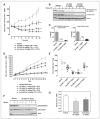
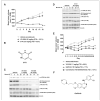
Dysregulation of the PI3K/mTOR pathway, either through amplifications, deletions, or as a direct result of mutations, has been closely linked to the development and progression of a wide range of cancers. Moreover, this pathway activation is a poor prognostic marker for many tumor types and confers resistance to various cancer therapies. Here, we describe VS-5584, a novel, low-molecular weight compound with equivalent potent activity against mTOR (IC(50) = 37 nmol/L) and all class I phosphoinositide 3-kinase (PI3K) isoforms IC(50): PI3Kα = 16 nmol/L; PI3Kβ = 68 nmol/L; PI3Kγ = 25 nmol/L; PI3Kδ = 42 nmol/L, without relevant activity on 400 lipid and protein kinases. VS-5584 shows robust modulation of cellular PI3K/mTOR pathways, inhibiting phosphorylation of substrates downstream of PI3K and mTORC1/2. A large human cancer cell line panel screen (436 lines) revealed broad antiproliferative sensitivity and that cells harboring mutations in PI3KCA are generally more sensitive toward VS-5584 treatment. VS-5584 exhibits favorable pharmacokinetic properties after oral dosing in mice and is well tolerated. VS-5584 induces long-lasting and dose-dependent inhibition of PI3K/mTOR signaling in tumor tissue, leading to tumor growth inhibition in various rapalog-sensitive and -resistant human xenograft models. Furthermore, VS-5584 is synergistic with an EGF receptor inhibitor in a gastric tumor model. The unique selectivity profile and favorable pharmacologic and pharmaceutical properties of VS-5584 and its efficacy in a wide range of human tumor models supports further investigations of VS-5584 in clinical trials.
©2012 AACR.
Figures

References
-
- Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B. The emerging mechanisms of isoform-specific PI3K signalling. Nat Rev Mol Cell Biol. 2010;11:329–41. - PubMed
-
- Engelman JA. Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer. 2009;9:550–62. - PubMed
-
- Huang S, Bjornsti MA, Houghton PJ. Rapamycins: mechanism of action and cellular resistance. Cancer Biol Ther. 2003;2:222–32. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

