GATA-6 and NF-κB activate CPI-17 gene transcription and regulate Ca2+ sensitization of smooth muscle contraction
- PMID: 23275439
- PMCID: PMC3623078
- DOI: 10.1128/MCB.00626-12
GATA-6 and NF-κB activate CPI-17 gene transcription and regulate Ca2+ sensitization of smooth muscle contraction
Abstract
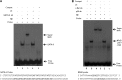
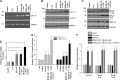
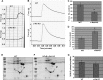
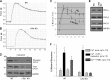
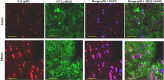
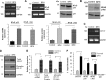
Protein kinase C (PKC)-potentiated inhibitory protein of 17 kDa (CPI-17) inhibits myosin light chain phosphatase, altering the levels of myosin light chain phosphorylation and Ca(2+) sensitivity in smooth muscle. In this study, we characterized the CPI-17 promoter and identified binding sites for GATA-6 and nuclear factor kappa B (NF-κB). GATA-6 and NF-κB upregulated CPI-17 expression in cultured human and mouse bladder smooth muscle (BSM) cells in an additive manner. CPI-17 expression was decreased upon GATA-6 silencing in cultured BSM cells and in BSM from NF-κB knockout (KO) mice. Moreover, force maintenance by BSM strips from KO mice was decreased compared with the force maintenance of BSM strips from wild-type mice. GATA-6 and NF-κB overexpression was associated with CPI-17 overexpression in BSM from men with benign prostatic hyperplasia (BPH)-induced bladder hypertrophy and in a mouse model of bladder outlet obstruction. Thus, aberrant expression of NF-κB and GATA-6 deregulates CPI-17 expression and the contractile function of smooth muscle. Our data provide insight into how GATA-6 and NF-κB mediate CPI-17 transcription, PKC-mediated signaling, and BSM remodeling associated with lower urinary tract symptoms in patients with BPH.
Figures








Similar articles
-
Mechanical stretch upregulates proteins involved in Ca2+ sensitization in urinary bladder smooth muscle hypertrophy.Am J Physiol Cell Physiol. 2014 Sep 15;307(6):C542-53. doi: 10.1152/ajpcell.00033.2014. Epub 2014 Jul 16. Am J Physiol Cell Physiol. 2014. PMID: 25031021 Free PMC article.
-
NF-κB and GATA-Binding Factor 6 Repress Transcription of Caveolins in Bladder Smooth Muscle Hypertrophy.Am J Pathol. 2019 Apr;189(4):847-867. doi: 10.1016/j.ajpath.2018.12.013. Epub 2019 Jan 30. Am J Pathol. 2019. PMID: 30707892 Free PMC article.
-
Transcriptional repression of Caveolin-1 (CAV1) gene expression by GATA-6 in bladder smooth muscle hypertrophy in mice and human beings.Am J Pathol. 2011 May;178(5):2236-51. doi: 10.1016/j.ajpath.2011.01.038. Am J Pathol. 2011. PMID: 21514437 Free PMC article.
-
Mechanisms underlying the pathogenesis of hyper-contractility of bronchial smooth muscle in allergic asthma.J Smooth Muscle Res. 2017;53(0):37-47. doi: 10.1540/jsmr.53.37. J Smooth Muscle Res. 2017. PMID: 28484126 Free PMC article. Review.
-
Protein kinase network in the regulation of phosphorylation and dephosphorylation of smooth muscle myosin light chain.Mol Cell Biochem. 2003 Jun;248(1-2):105-14. doi: 10.1023/a:1024180101032. Mol Cell Biochem. 2003. PMID: 12870661 Review.
Cited by
-
Regulator of G protein signaling 4 is a novel target of GATA-6 transcription factor.Biochem Biophys Res Commun. 2017 Feb 12;483(3):923-929. doi: 10.1016/j.bbrc.2016.10.024. Epub 2016 Oct 13. Biochem Biophys Res Commun. 2017. PMID: 27746176 Free PMC article.
-
Viral mediated knockdown of GATA6 in SMA iPSC-derived astrocytes prevents motor neuron loss and microglial activation.Glia. 2022 May;70(5):989-1004. doi: 10.1002/glia.24153. Epub 2022 Jan 28. Glia. 2022. PMID: 35088910 Free PMC article.
-
The retinoblastoma tumor suppressor modulates DNA repair and radioresponsiveness.Clin Cancer Res. 2014 Nov 1;20(21):5468-5482. doi: 10.1158/1078-0432.CCR-14-0326. Epub 2014 Aug 27. Clin Cancer Res. 2014. PMID: 25165096 Free PMC article.
-
NF-кB c-Rel modulates pre-fibrotic changes in human fibroblasts.Arch Dermatol Res. 2022 Dec;314(10):943-951. doi: 10.1007/s00403-021-02310-2. Epub 2021 Dec 9. Arch Dermatol Res. 2022. PMID: 34888734 Free PMC article.
-
Mechanical stretch upregulates proteins involved in Ca2+ sensitization in urinary bladder smooth muscle hypertrophy.Am J Physiol Cell Physiol. 2014 Sep 15;307(6):C542-53. doi: 10.1152/ajpcell.00033.2014. Epub 2014 Jul 16. Am J Physiol Cell Physiol. 2014. PMID: 25031021 Free PMC article.
References
-
- Adelstein RS, Eisenberg E. 1980. Regulation and kinetics of the actin-myosin-ATP interaction. Annu. Rev. Biochem. 49: 921– 956 - PubMed
-
- Somlyo AP, Somlyo AV. 2003. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: modulated by G proteins, kinases, and myosin phosphatase. Physiol. Rev. 83: 1325– 1358 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
