Ragweed pollen extract intensifies lipopolysaccharide-induced priming of NLRP3 inflammasome in human macrophages
- PMID: 23278511
- PMCID: PMC3719949
- DOI: 10.1111/imm.12052
Ragweed pollen extract intensifies lipopolysaccharide-induced priming of NLRP3 inflammasome in human macrophages
Abstract
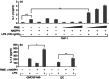
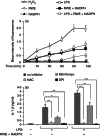
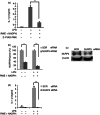
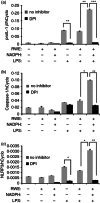
Ragweed pollen extract (RWE) possesses intrinsic NADPH oxidase activity that induces oxidative stress by initiating the production of intracellular reactive oxygen species (ROS). The ROS are important contributors to the manifestation of allergic inflammation; furthermore, concomitant exposure to an allergen and an endotoxin trigger a stronger inflammatory response. One of the main pro-inflammatory cytokines produced in inflammatory responses is interleukin-1β (IL-1β), and its production is associated with caspase-1-containing inflammasome complexes. Intracellular ROS have been implicated in NLRP3 inflammasome-mediated IL-1β production, therefore, we aimed to study whether RWE influences the function of NLRP3 inflammasome. Here we describe that, in the presence of NADPH, RWE significantly elevates lipopolysaccharide-induced IL-1β production of THP-1 cells as well as human primary macrophages and dendritic cells. We also demonstrate that increased IL-1β production is mediated through NLRP3 inflammasome in THP-1 macrophages. We provide evidence that RWE elevates cytosolic ROS level in these cells, and ROS inhibitors abolish IL-1β production. Furthermore, we show that RWE enhances lipopolysaccharide-induced gene transcription/expression of pro-IL-1β and key components of the inflammasome via a ROS-dependent mechanism.
© 2012 University of Debrecen Immunology © 2012 Blackwell Publishing Ltd.
Figures


Similar articles
-
Mechanisms that lead to the regulation of NLRP3 inflammasome expression and activation in human dental pulp fibroblasts.Mol Immunol. 2015 Aug;66(2):253-62. doi: 10.1016/j.molimm.2015.03.009. Epub 2015 Apr 2. Mol Immunol. 2015. PMID: 25863775
-
Aloe vera downregulates LPS-induced inflammatory cytokine production and expression of NLRP3 inflammasome in human macrophages.Mol Immunol. 2013 Dec;56(4):471-9. doi: 10.1016/j.molimm.2013.05.005. Epub 2013 Aug 1. Mol Immunol. 2013. PMID: 23911403
-
Electronegative LDL induces priming and inflammasome activation leading to IL-1β release in human monocytes and macrophages.Biochim Biophys Acta. 2015 Nov;1851(11):1442-9. doi: 10.1016/j.bbalip.2015.08.009. Epub 2015 Aug 29. Biochim Biophys Acta. 2015. PMID: 26327597
-
Mechanism and Regulation of NLRP3 Inflammasome Activation.Trends Biochem Sci. 2016 Dec;41(12):1012-1021. doi: 10.1016/j.tibs.2016.09.002. Epub 2016 Sep 23. Trends Biochem Sci. 2016. PMID: 27669650 Free PMC article. Review.
-
The NLRP3 inflammasome in health and disease: the good, the bad and the ugly.Clin Exp Immunol. 2011 Oct;166(1):1-15. doi: 10.1111/j.1365-2249.2011.04440.x. Epub 2011 Jul 15. Clin Exp Immunol. 2011. PMID: 21762124 Free PMC article. Review.
Cited by
-
Birch Pollen Induces Toll-Like Receptor 4-Dependent Dendritic Cell Activation Favoring T Cell Responses.Front Allergy. 2021 Aug 12;2:680937. doi: 10.3389/falgy.2021.680937. eCollection 2021. Front Allergy. 2021. PMID: 35386993 Free PMC article.
-
Overview of the Mechanisms of Oxidative Stress: Impact in Inflammation of the Airway Diseases.Antioxidants (Basel). 2022 Nov 13;11(11):2237. doi: 10.3390/antiox11112237. Antioxidants (Basel). 2022. PMID: 36421423 Free PMC article. Review.
-
Radiation-Detoxified Form of Endotoxin Effectively Activates Th1 Responses and Attenuates Ragweed-Induced Th2-Type Airway Inflammation in Mice.Int J Mol Sci. 2024 Jan 27;25(3):1581. doi: 10.3390/ijms25031581. Int J Mol Sci. 2024. PMID: 38338861 Free PMC article.
-
Natural Compounds as Regulators of NLRP3 Inflammasome-Mediated IL-1β Production.Mediators Inflamm. 2016;2016:5460302. doi: 10.1155/2016/5460302. Epub 2016 Sep 8. Mediators Inflamm. 2016. PMID: 27672241 Free PMC article. Review.
-
The activation of pyrin domain-containing-3 inflammasome depends on lipopolysaccharide from Porphyromonas gingivalis and extracellular adenosine triphosphate in cultured oral epithelial cells.BMC Oral Health. 2015 Oct 29;15(1):133. doi: 10.1186/s12903-015-0115-6. BMC Oral Health. 2015. PMID: 26511096 Free PMC article.
References
-
- Dharajiya NG, Bacsi A, Boldogh I, Sur S. Pollen NAD(P)H oxidases and their contribution to allergic inflammation. Immunol Allergy Clin North Am. 2007;27:45–63. - PubMed
-
- Reed CE, Milton DK. Endotoxin-stimulated innate immunity: a contributing factor for asthma. J Allergy Clin Immunol. 2001;108:157–66. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

