Effects of digesting chondroitin sulfate proteoglycans on plasticity in cat primary visual cortex
- PMID: 23283337
- PMCID: PMC6618616
- DOI: 10.1523/JNEUROSCI.2283-12.2013
Effects of digesting chondroitin sulfate proteoglycans on plasticity in cat primary visual cortex
Abstract
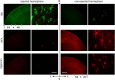
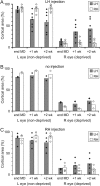
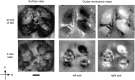
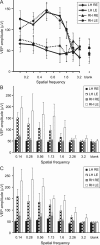
Monocular deprivation (MD) during a critical period of postnatal development produces significant changes in the anatomy and physiology of the visual cortex, and the deprived eye becomes amblyopic. Extracellular matrix molecules have a major role in restricting plasticity such that the ability to recover from MD decreases with age. Chondroitin sulfate proteoglycans (CSPGs) act as barriers to cell migration and axon growth. Previous studies showing that degradation of CSPGs by the bacterial enzyme chondroitinase can restore plasticity in the adult rat visual cortex suggest a potential treatment for amblyopia. Here MD was imposed in cats from the start of the critical period until 3.5 months of age. The deprived eye was reopened, the functional architecture of the visual cortex was assessed by optical imaging of intrinsic signals, and chondroitinase was injected into one hemisphere. Imaging was repeated 1 and 2 weeks postinjection, and visually evoked potentials (VEPs) and single-cell activity were recorded. Immunohistochemistry showed that digestion of CSPGs had been successful. After 2 weeks of binocular exposure, some recovery of deprived-eye responses occurred when chondroitinase had been injected into the hemisphere contralateral to that eye; when injected into the ipsilateral hemisphere, no recovery was seen. Deprived-eye VEPs were no larger in the injected hemisphere than in the opposite hemisphere. The small number of neurons dominated by the deprived eye exhibited poor tuning characteristics. These results suggest that despite structural effects of chondroitinase in adult cat V1, plasticity was not sufficiently restored to enable significant functional recovery of the deprived eye.
Figures



References
-
- Attwell D, Laughlin SB. An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab. 2001;21:1133–1145. - PubMed
-
- Baro JA, Lehmkuhle S, Kratz KE. Electroretinograms and visual evoked potentials in long-term monocularly deprived cats. Invest Ophthalmol Vis Sci. 1990;31:1405–1409. - PubMed
-
- Bear MF, Singer W. Modulation of visual cortical plasticity by acetylcholine and noradrenaline. Nature. 1986;320:172–176. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
