Angiotensin receptor agonistic autoantibody-mediated soluble fms-like tyrosine kinase-1 induction contributes to impaired adrenal vasculature and decreased aldosterone production in preeclampsia
- PMID: 23283357
- PMCID: PMC4131732
- DOI: 10.1161/HYPERTENSIONAHA.111.00157
Angiotensin receptor agonistic autoantibody-mediated soluble fms-like tyrosine kinase-1 induction contributes to impaired adrenal vasculature and decreased aldosterone production in preeclampsia
Erratum in
- Hypertension. 2013 Jun;61(6):e61
Abstract
Preeclampsia (PE) is a life-threatening hypertensive disorder during pregnancy associated with decreased circulating aldosterone levels. However, the molecular mechanisms underlying aldosterone reduction in PE remain unidentified. Here we demonstrate that reduced circulating aldosterone levels in preeclamptic women are associated with the presence of angiotensin II type 1 receptor agonistic autoantibody and elevated soluble Fms-like tyrosine kinase-1, 2 prominent pathogenic factors in PE. Using an adoptive transfer animal model of PE, we provide in vivo evidence that the injection of IgG from women with PE, but not IgG from normotensive individuals, resulted in hypertension, proteinuria, and a reduction in aldosterone production from 1377 ± 272 pg/mL to 544 ± 92 pg/mL (P<0.05) in pregnant mice. These features were prevented by coinjection with an epitope peptide that blocks antibody-mediated angiotensin type 1 receptor activation. In contrast, injection of IgG from preeclamptic women into nonpregnant mice induced aldosterone levels from 213 ± 24 pg/mL to 615 ± 48 pg/mL (P<0.05). These results indicate that maternal circulating autoantibody in preeclamptic women is a detrimental factor causing decreased aldosterone production via angiotensin type 1 receptor activation in a pregnancy-dependent manner. Next, we found that circulating soluble Fms-like tyrosine kinase-1 was only induced in autoantibody-injected pregnant mice but not nonpregnant mice. As such, we further observed vascular impairment in adrenal glands of pregnant mice. Finally, we demonstrated that infusion of vascular endothelial growth factor(121) attenuated autoantibody-induced adrenal gland vascular impairment resulting in a recovery in circulating aldosterone (from 544 ± 92 to 1110 ± 269 pg/mL; P<0.05). Overall, we revealed that angiotensin II type 1 receptor agonistic autoantibody-induced soluble Fms-like tyrosine kinase-1 elevation is a novel pathogenic mechanism underlying decreased aldosterone production in PE.
Figures
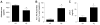
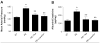
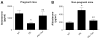

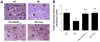
Similar articles
-
Autoantibody-mediated complement C3a receptor activation contributes to the pathogenesis of preeclampsia.Hypertension. 2012 Sep;60(3):712-21. doi: 10.1161/HYPERTENSIONAHA.112.191817. Epub 2012 Aug 6. Hypertension. 2012. PMID: 22868393 Free PMC article.
-
Angiotensin receptor agonistic autoantibody-mediated tumor necrosis factor-alpha induction contributes to increased soluble endoglin production in preeclampsia.Circulation. 2010 Jan 26;121(3):436-44. doi: 10.1161/CIRCULATIONAHA.109.902890. Epub 2010 Jan 11. Circulation. 2010. PMID: 20065159 Free PMC article.
-
Autoantibody-mediated angiotensin receptor activation contributes to preeclampsia through tumor necrosis factor-alpha signaling.Hypertension. 2010 May;55(5):1246-53. doi: 10.1161/HYPERTENSIONAHA.110.150540. Epub 2010 Mar 29. Hypertension. 2010. PMID: 20351341 Free PMC article.
-
Dysregulation of anti-angiogenic agents (sFlt-1, PLGF, and sEndoglin) in preeclampsia--a step forward but not the definitive answer.J Reprod Immunol. 2009 Nov;82(2):106-11. doi: 10.1016/j.jri.2009.09.001. Epub 2009 Oct 23. J Reprod Immunol. 2009. PMID: 19853925 Review.
-
The renin-angiotensin-aldosterone system in pre-eclampsia: the delicate balance between good and bad.Clin Sci (Lond). 2014 Apr;126(8):537-44. doi: 10.1042/CS20130455. Clin Sci (Lond). 2014. PMID: 24400721 Review.
Cited by
-
Vascular mechanisms and molecular targets in hypertensive pregnancy and preeclampsia.Am J Physiol Heart Circ Physiol. 2020 Sep 1;319(3):H661-H681. doi: 10.1152/ajpheart.00202.2020. Epub 2020 Aug 7. Am J Physiol Heart Circ Physiol. 2020. PMID: 32762557 Free PMC article. Review.
-
Recent advances in central cardiovascular control: sex, ROS, gas and inflammation.F1000Res. 2016 Mar 31;5:F1000 Faculty Rev-420. doi: 10.12688/f1000research.7987.1. eCollection 2016. F1000Res. 2016. PMID: 27092251 Free PMC article. Review.
-
Combined Effects of Gestational Phthalate Exposure and Zinc Deficiency on Steroid Metabolism and Growth.Toxicol Sci. 2017 Apr 1;156(2):469-479. doi: 10.1093/toxsci/kfx008. Toxicol Sci. 2017. PMID: 28115639 Free PMC article.
-
Regulation of vascular angiotensin II type 1 and type 2 receptor and angiotensin-(1-7)/MasR signaling in normal and hypertensive pregnancy.Biochem Pharmacol. 2024 Feb;220:115963. doi: 10.1016/j.bcp.2023.115963. Epub 2023 Dec 5. Biochem Pharmacol. 2024. PMID: 38061417 Free PMC article. Review.
-
Role of agonistic autoantibodies to the angiotensin II type 1 receptor (AT1-AA) in pathogenesis of preeclampsia.Glob Med Genet. 2025 Feb 6;12(2):100041. doi: 10.1016/j.gmg.2025.100041. eCollection 2025 Jun. Glob Med Genet. 2025. PMID: 40027241 Free PMC article. Review.
References
-
- Roberts JM, Pearson G, Cutler J, Lindheimer M. Summary of the nhlbi working group on research on hypertension during pregnancy. Hypertension. 2003;41:437–445. - PubMed
-
- Sibai B, Dekker G, Kupferminc M. Pre-eclampsia. Lancet. 2005;365:785–799. - PubMed
-
- Lindheimer MD, Umans JG. Explaining and predicting preeclampsia. N Engl J Med. 2006;355:1056–1058. - PubMed
-
- Young BC, Levine RJ, Karumanchi SA. Pathogenesis of preeclampsia. Annu Rev Pathol. 5:173–192. - PubMed
-
- Maynard SE, Min JY, Merchan J, Lim KH, Li J, Mondal S, Libermann TA, Morgan JP, Sellke FW, Stillman IE, Epstein FH, Sukhatme VP, Karumanchi SA. Excess placental soluble fms-like tyrosine kinase 1 (sflt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest. 2003;111:649–658. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

