Pyrosequencing reveals the influence of organic and conventional farming systems on bacterial communities
- PMID: 23284808
- PMCID: PMC3526490
- DOI: 10.1371/journal.pone.0051897
Pyrosequencing reveals the influence of organic and conventional farming systems on bacterial communities
Abstract
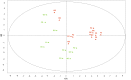
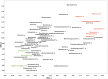
It has been debated how different farming systems influence the composition of soil bacterial communities, which are crucial for maintaining soil health. In this research, we applied high-throughput pyrosequencing of V1 to V3 regions of bacterial 16S rRNA genes to gain further insight into how organic and conventional farming systems and crop rotation influence bulk soil bacterial communities. A 2×2 factorial experiment consisted of two agriculture management systems (organic versus conventional) and two crop rotations (flax-oat-fababean-wheat versus flax-alfalfa-alfalfa-wheat) was conducted at the Glenlea Long-Term Crop Rotation and Management Station, which is Canada's oldest organic-conventional management study field. Results revealed that there is a significant difference in the composition of bacterial genera between organic and conventional management systems but crop rotation was not a discriminator factor. Organic farming was associated with higher relative abundance of Proteobacteria, while Actinobacteria and Chloroflexi were more abundant in conventional farming. The dominant genera including Blastococcus, Microlunatus, Pseudonocardia, Solirubrobacter, Brevundimonas, Pseudomonas, and Stenotrophomonas exhibited significant variation between the organic and conventional farming systems. The relative abundance of bacterial communities at the phylum and class level was correlated to soil pH rather than other edaphic properties. In addition, it was found that Proteobacteria and Actinobacteria were more sensitive to pH variation.
Conflict of interest statement
Figures

References
-
- Doran JW, Parkin TB (1994) Defining and assessing soil quality; Doran JW, Coleman DC, Bezdicek DF, Stewart BA, editors. Madison, Wisconsin USA: Soil Science Society of America, America Society of Agronomy. pp 3–21.
-
- van Bruggen AHC, Semenov AM (2000) In search of biological indicators for soil health and disease suppression. Appl Soil Ecol 15: 13–24.
-
- Sprent JI (1979) The biology of nitrogen-fixing organisms. London; New York: McGraw-Hill. 196 p.
-
- Dorn E, Hellwig M, Reineke W, Knackmuss HJ (1974) Isolation and characterization of a 3-chlorobenzoate degrading pseudomonad. Arch Microbiol 99: 61–70. - PubMed
-
- Torstensson L (1980) Role of microorganisms in decomposition. In: Hance RJ, editor. Interaction between hercibides and the soil London: Academic Press. 159–177.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

