Increased Tumor Necrosis Factor (TNF)-α and its promoter polymorphisms correlate with disease progression and higher susceptibility towards vitiligo
- PMID: 23284977
- PMCID: PMC3527546
- DOI: 10.1371/journal.pone.0052298
Increased Tumor Necrosis Factor (TNF)-α and its promoter polymorphisms correlate with disease progression and higher susceptibility towards vitiligo
Abstract
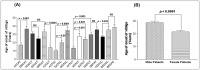
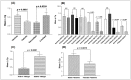
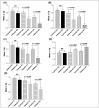
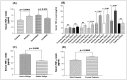
Tumor Necrosis Factor (TNF)-α, is a paracrine inhibitor of melanocytes, which plays a critical role in the pathogenesis of several autoimmune diseases including vitiligo, as abnormal immune responses have frequently been observed in vitiligo patients. Moreover, vitiligo patients show higher lesion levels of TNF-α. Genetic polymorphisms in the promoter region of TNF-α are involved in the regulation of its expression. The present study explores TNF-α promoter polymorphisms and correlates them with TNF-α transcript and protein levels in vitiligo patients and controls of Gujarat along with its effect on disease onset and progression. PCR-RFLP technique was used for genotyping of these polymorphisms in 977 vitiligo patients and 990 controls. TNF-α transcript and protein levels were measured by Real time PCR and ELISA respectively. The genotype and allele frequencies for the investigated polymorphisms were significantly associated with vitiligo patients. The study revealed significant increase in TNF-α transcript and protein levels in vitiligo patients compared to controls. In particular, haplotypes: AATCC, AACCT, AGTCT, GATCT, GATCC and AGCCT were found to increase the TNF-α levels in vitiligo patients. Analysis of TNF-α levels based on the gender and disease progression suggests that female patients and patients with active vitiligo had higher levels of TNF-α. Also, the TNF-α levels were high in patients with generalized vitiligo as compared to localized vitiligo. Age of onset analysis of the disease suggests that the haplotypes: AACAT, AACCT, AATCC and AATCT had a profound effect in the early onset of the disease. Moreover, the analysis suggests that female patients had an early onset of vitiligo. Overall, our results suggest that TNF-α promoter polymorphisms may be genetic risk factors for susceptibility and progression of the disease. The up-regulation of TNF-α transcript and protein levels in individuals with susceptible haplotypes advocates the crucial role of TNF-α in autoimmune pathogenesis of vitiligo.
Figures
References
-
- Taieb A, Picardo M (2009) Vitiligo. N. Engl. J. Med. 360: 160–169. - PubMed
-
- Spritz RA (2008) The genetics of generalized vitiligo. Curr. Dir. Autoimmun. 10: 244–257. - PubMed
-
- Das SK, Majumder PP, Chakraborty R, Majumdar TK, Haldar B (1985) Studies on vitiligo: Epidemiological profile in Calcutta, India. Genet. Epidemiol. 2: 71–78. - PubMed
-
- Alkhateeb A, Fain PR, Thody A, Bennett DC, Spritz RA (2003) Epidemiology of vitiligo and associated autoimmune diseases in Caucasian probands and their relatives. Pigment Cell Res. 16: 208–214. - PubMed
-
- Laberge G, Mailloux CM, Gowan K, Holland P, Bennett DC, et al. (2005) Early disease onset and increased risk of other autoimmune diseases in familial generalized vitiligo. Pigment Cell Res. 18: 300–305. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

