Structural and functional similarities of calcium homeostasis modulator 1 (CALHM1) ion channel with connexins, pannexins, and innexins
- PMID: 23300080
- PMCID: PMC3585051
- DOI: 10.1074/jbc.M112.409789
Structural and functional similarities of calcium homeostasis modulator 1 (CALHM1) ion channel with connexins, pannexins, and innexins
Abstract
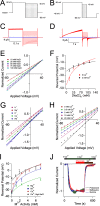
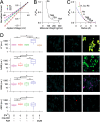
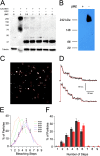
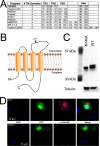
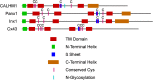
CALHM1 (calcium homeostasis modulator 1) forms a plasma membrane ion channel that mediates neuronal excitability in response to changes in extracellular Ca(2+) concentration. Six human CALHM homologs exist with no homology to other proteins, although CALHM1 is conserved across >20 species. Here we demonstrate that CALHM1 shares functional and quaternary and secondary structural similarities with connexins and evolutionarily distinct innexins and their vertebrate pannexin homologs. A CALHM1 channel is a hexamer, comprised of six monomers, each of which possesses four transmembrane domains, cytoplasmic amino and carboxyl termini, an amino-terminal helix, and conserved extracellular cysteines. The estimated pore diameter of the CALHM1 channel is ∼14 Å, enabling permeation of large charged molecules. Thus, CALHMs, connexins, and pannexins and innexins are structurally related protein families with shared and distinct functional properties.
Figures
Similar articles
-
Calcium homeostasis modulator (CALHM) ion channels.Pflugers Arch. 2016 Mar;468(3):395-403. doi: 10.1007/s00424-015-1757-6. Epub 2015 Nov 25. Pflugers Arch. 2016. PMID: 26603282 Free PMC article. Review.
-
A mechanism of CALHM1 ion channel gating.Am J Physiol Cell Physiol. 2025 Apr 1;328(4):C1109-C1124. doi: 10.1152/ajpcell.00925.2024. Epub 2025 Feb 21. Am J Physiol Cell Physiol. 2025. PMID: 39981825 Free PMC article.
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
-
Calcium homeostasis modulator 1 (CALHM1) is the pore-forming subunit of an ion channel that mediates extracellular Ca2+ regulation of neuronal excitability.Proc Natl Acad Sci U S A. 2012 Jul 10;109(28):E1963-71. doi: 10.1073/pnas.1204023109. Epub 2012 Jun 18. Proc Natl Acad Sci U S A. 2012. PMID: 22711817 Free PMC article.
-
Phylogenetic and bioinformatic analysis of gap junction-related proteins, innexins, pannexins and connexins.Biomed Res. 2010 Apr;31(2):133-42. doi: 10.2220/biomedres.31.133. Biomed Res. 2010. PMID: 20460741
Cited by
-
SWELL1, a plasma membrane protein, is an essential component of volume-regulated anion channel.Cell. 2014 Apr 10;157(2):447-458. doi: 10.1016/j.cell.2014.03.024. Cell. 2014. PMID: 24725410 Free PMC article.
-
Intrinsic properties and regulation of Pannexin 1 channel.Channels (Austin). 2014;8(2):103-9. doi: 10.4161/chan.27545. Epub 2014 Jan 13. Channels (Austin). 2014. PMID: 24419036 Free PMC article. Review.
-
Expression and Localization of Connexins in the Outer Retina of the Mouse.J Mol Neurosci. 2016 Feb;58(2):178-92. doi: 10.1007/s12031-015-0654-y. Epub 2015 Oct 9. J Mol Neurosci. 2016. PMID: 26453550
-
Cryo-EM structures of calcium homeostasis modulator channels in diverse oligomeric assemblies.Sci Adv. 2020 Jul 17;6(29):eaba8105. doi: 10.1126/sciadv.aba8105. eCollection 2020 Jul. Sci Adv. 2020. PMID: 32832629 Free PMC article.
-
Structural studies of N-terminal mutants of Connexin 26 and Connexin 32 using (1)H NMR spectroscopy.Arch Biochem Biophys. 2016 Oct 15;608:8-19. doi: 10.1016/j.abb.2016.06.019. Epub 2016 Jul 1. Arch Biochem Biophys. 2016. PMID: 27378082 Free PMC article.
References
-
- Dreses-Werringloer U., Lambert J. C., Vingtdeux V., Zhao H., Vais H., Siebert A., Jain A., Koppel J., Rovelet-Lecrux A., Hannequin D., Pasquier F., Galimberti D., Scarpini E., Mann D., Lendon C., Campion D., Amouyel P., Davies P., Foskett J. K., Campagne F., Marambaud P. (2008) A polymorphism in CALHM1 influences Ca2+ homeostasis, Aβ levels, and Alzheimer's disease risk. Cell 133, 1149–1161 - PMC - PubMed
-
- Lambert J. C., Amouyel P. (2007) Genetic heterogeneity of Alzheimer's disease. Complexity and advances. Psychoneuroendocrinology 32, S62–S70 - PubMed
-
- Ma Z., Siebert A. P., Cheung K. H., Lee R. J., Johnson B., Cohen A. S., Vingtdeux V., Marambaud P., Foskett J. K. (2012) Calcium homeostasis modulator 1 (CALHM1) is the pore-forming subunit of an ion channel that mediates extracellular Ca2+ regulation of neuronal excitability. Proc. Natl. Acad. Sci. U.S.A. 109, E1963–E1971 - PMC - PubMed
-
- Gallego-Sandín S., Alonso M. T., García-Sancho J. (2011) Calcium homoeostasis modulator 1 (CALHM1) reduces the calcium content of the endoplasmic reticulum (ER) and triggers ER stress. Biochem. J. 437, 469–475 - PubMed
-
- Moreno-Ortega A. J., Ruíz-Nuño A., García A. G., Cano-Abad M. F. (2010) Mitochondria sense with different kinetics the calcium entering into HeLa cells through calcium channels CALHM1 and mutated P86L-CALHM1. Biochem. Biophys. Res. Commun. 391, 722–726 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

