Successful adjuvant bi-weekly gemcitabine chemotherapy for pancreatic cancer without impairing patients' quality of life
- PMID: 23302293
- PMCID: PMC3548728
- DOI: 10.1186/1477-7819-11-3
Successful adjuvant bi-weekly gemcitabine chemotherapy for pancreatic cancer without impairing patients' quality of life
Abstract
Background: Although adjuvant gemcitabine (GEM) chemotherapy for pancreatic cancer is standard, the quality of life (QOL) in those patients is still impaired by the standard regimen of GEM. Therefore, we studied whether mild dose-intensity adjuvant chemotherapy with bi-weekly GEM administration could provide a survival benefit with acceptable QOL to the patients with pancreatic cancer.
Methods: After a phase I trial, an adjuvant bi-weekly 1,000 mg/m2 of GEM chemotherapy was performed in 58 patients with pancreatic cancer for at least 12 courses (Group A). In contrast, 36 patients who declined the adjuvant bi-weekly GEM chemotherapy underwent traditional adjuvant 5FU-based chemotherapy (Group B). Careful periodical follow-ups for side effects of GEM and disease recurrence, and assessment of patients' QOL using the EORTC QOL questionnaire (QLQ-C30) and pancreatic cancer-specific supplemental module (QLQ-PAN26) were performed. Retrospectively, the degree of side effects, patients' QOL, compliance rate, disease-free survival (DFS), and overall survival (OS) in Group A were compared with those in Group B.
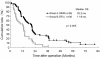
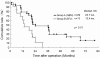
Results: No severe side effects (higher than Grade 2 according to the common toxicity criteria of ECOG) were observed, except for patients in Group B, who were switched to the standard GEM chemotherapy. Patients' QOL was better in Group A than B (fatigue: 48.9 ± 32.1 versus 68.1 ± 36.3, nausea and vomiting: 26.8 ± 20.4 versus 53.7 ± 32.6, diarrhea: 21.0 ± 22.6 versus 53.9 ± 38.5, difficulty gaining weight: 49.5 ± 34.4 versus 67.7 ± 40.5, P < 0.05). Compliance rates in Groups A and B were 93% and 47%. There was a significant difference in the median DFS between both groups (Group A : B =12.5 : 6.6 months, P < 0.001). The median OS of Group A was prolonged markedly compared with Group B (20.2 versus 11.9 months, P < 0.005). For OS between both groups, univariate analysis revealed no statistical difference in 69-year-old or under females, and T1-2 factors, moreover, multivariate analysis indicated three factors, such as bi-weekly adjuvant GEM chemotherapy, T2 or less, and R0.
Conclusions: Adjuvant chemotherapy with bi-weekly GEM offered not only the advantage of survival benefits but the excellent compliance with acceptable QOL for postoperative pancreatic cancer patients.
Figures


Similar articles
-
Postoperative adjuvant gemcitabine and concurrent radiation after curative resection of pancreatic head carcinoma: a phase II study.Int J Radiat Oncol Biol Phys. 2003 Jul 15;56(4):974-80. doi: 10.1016/s0360-3016(03)00164-0. Int J Radiat Oncol Biol Phys. 2003. PMID: 12829132 Clinical Trial.
-
Phase 2 multi-institutional trial evaluating gemcitabine and stereotactic body radiotherapy for patients with locally advanced unresectable pancreatic adenocarcinoma.Cancer. 2015 Apr 1;121(7):1128-37. doi: 10.1002/cncr.29161. Epub 2014 Dec 23. Cancer. 2015. PMID: 25538019 Free PMC article. Clinical Trial.
-
Survival after chemoradiation in resected pancreatic cancer: the impact of adjuvant gemcitabine.Int J Radiat Oncol Biol Phys. 2012 Jul 1;83(3):e331-5. doi: 10.1016/j.ijrobp.2012.01.008. Epub 2012 Mar 13. Int J Radiat Oncol Biol Phys. 2012. PMID: 22420967
-
Gemcitabine and S-1 combination chemotherapy versus gemcitabine alone for locally advanced and metastatic pancreatic cancer: a meta-analysis of randomized controlled trials in Asia.J Chemother. 2015 Aug;27(4):227-34. doi: 10.1179/1973947815Y.0000000013. Epub 2015 Mar 20. J Chemother. 2015. PMID: 25790948
-
Gemcitabine-based combination therapy compared with gemcitabine alone for advanced pancreatic cancer: a meta-analysis of nine randomized controlled trials.Hepatobiliary Pancreat Dis Int. 2017 Jun;16(3):236-244. doi: 10.1016/s1499-3872(17)60022-5. Hepatobiliary Pancreat Dis Int. 2017. PMID: 28603091 Review.
Cited by
-
Landscape of Health-Related Quality of Life in Patients With Early-Stage Pancreatic Cancer Receiving Adjuvant or Neoadjuvant Chemotherapy: A Systematic Literature Review.Pancreas. 2020 Mar;49(3):393-407. doi: 10.1097/MPA.0000000000001507. Pancreas. 2020. PMID: 32132518 Free PMC article.
-
Evaluation of host quality of life and immune function in breast cancer patients treated with combination of adjuvant chemotherapy and oral administration of Lentinula edodes mycelia extract.Onco Targets Ther. 2013 Jul 9;6:853-9. doi: 10.2147/OTT.S44169. Print 2013. Onco Targets Ther. 2013. PMID: 23874107 Free PMC article.
-
Management of Pancreatic Cancer and Its Microenvironment: Potential Impact of Nano-Targeting.Cancers (Basel). 2022 Jun 10;14(12):2879. doi: 10.3390/cancers14122879. Cancers (Basel). 2022. PMID: 35740545 Free PMC article. Review.
-
Linc-DYNC2H1-4 promotes EMT and CSC phenotypes by acting as a sponge of miR-145 in pancreatic cancer cells.Cell Death Dis. 2017 Jul 13;8(7):e2924. doi: 10.1038/cddis.2017.311. Cell Death Dis. 2017. PMID: 28703793 Free PMC article.
References
-
- Bakkevold KE, Amesiø B, Dahl O, Kambestad B. Adjuvant combination chemotherapy (AMF) following radical resection of carcinoma of the pancreas and pappilla of Vater–results of a controlled, prospective, randomised multicentre study. Eur J Cancer. 1993;29A:698–703. doi: 10.1016/S0959-8049(05)80349-1. - DOI - PubMed
-
- Burris HA III, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, Cripps MC, Portenoy RK, Storniolo AM, Tarassoff P, Nelson R, Dorr FA, Stephens CD, Von Hoff DD. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15:2403–2413. PMID: 9196156. - PubMed
-
- Lygidakis NJ, Berberabe AE, Spentzouris N, Dedemadi G, Kalligas T, Loukas G, Sotiropoulou V. A prospective randomized study using adjuvant locoregional chemoimmunotherapy in combination with surgery for pancreatic carcinoma. Hepatogastroenterology. 1998;45:2376–2381. - PubMed
-
- Klinkenbijl JH, Jeekel J, Sahmoud T, van Pel R, Couvreur ML, Veenhof CH, Arnaud JP, Gonzalez DG, de Wit LT, Hennipman A, Wils J. Adjuvant radiotherapy and 5-fluorouracil after curative resection of cancer of the pancreas and periampullary region: phase III trial of the EORTC gastrointestinal tract cancer cooperative group. Ann Surg. 1999;230:776–784. doi: 10.1097/00000658-199912000-00006. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

