Properties of two cataract-associated mutations located in the NH2 terminus of connexin 46
- PMID: 23302783
- PMCID: PMC3651606
- DOI: 10.1152/ajpcell.00344.2012
Properties of two cataract-associated mutations located in the NH2 terminus of connexin 46
Abstract
Mutations in connexin 46 are associated with congenital cataracts. The purpose of this project was to characterize cellular and functional properties of two congenital cataract-associated mutations located in the NH2 terminus of connexin 46: Cx46D3Y and Cx46L11S, which we found localized to gap junctional plaques like wild-type Cx46 in transfected HeLa cells. Dual two-microelectrode-voltage-clamp studies of Xenopus oocyte pairs injected with wild-type or mutant rat Cx46 showed that oocyte pairs injected with D3Y or L11S cRNA failed to induce gap junctional coupling, whereas oocyte pairs injected with Cx46 showed high levels of coupling. D3Y, but not L11S, functionally paired with wild-type Cx46. To determine whether coexpression of D3Y or L11S affected the junctional conductance produced by wild-type lens connexins, we studied pairs of oocytes coinjected with equal amounts of mutant and wild-type connexin cRNA. Expression of D3Y or L11S almost completely abolished gap junctional coupling induced by Cx46. In contrast, expression of D3Y or L11S failed to inhibit junctional conductance induced by Cx50. To examine effects of the D3Y and L11S mutations on hemichannel activity, hemichannel currents were measured in connexin cRNA-injected oocytes. Oocytes expressing D3Y exhibited reduced hemichannel activity as well as alterations in voltage gating and charge selectivity while oocytes expressing L11S showed no hemichannel activity. Moreover, coexpression of mutant with wild-type Cx50 or Cx46 gave rise to hemichannels with distinct electrophysiological properties, suggesting that the mutant connexins were forming heteromeric channels with wild-type connexins. These data suggest D3Y and L11S cause cataracts by similar but not identical mechanisms.
Keywords: cataract; connexin; gap junction; intercellular communication.
Figures
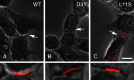
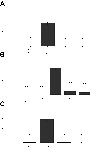






Comment in
-
Is half of a lens gap junction channel better than none? Focus on "properties of two cataract-associated mutations located in the NH2 terminus of connexin 46".Am J Physiol Cell Physiol. 2013 May 1;304(9):C821-2. doi: 10.1152/ajpcell.00035.2013. Epub 2013 Feb 7. Am J Physiol Cell Physiol. 2013. PMID: 23392114 No abstract available.
Similar articles
-
Characterization of a mouse Cx50 mutation associated with the No2 mouse cataract.Invest Ophthalmol Vis Sci. 1999 Jul;40(8):1844-50. Invest Ophthalmol Vis Sci. 1999. PMID: 10393059
-
Functional effects of Cx50 mutations associated with congenital cataracts.Am J Physiol Cell Physiol. 2014 Feb 1;306(3):C212-20. doi: 10.1152/ajpcell.00098.2013. Epub 2013 Sep 4. Am J Physiol Cell Physiol. 2014. PMID: 24005045 Free PMC article.
-
The cataract-inducing S50P mutation in Cx50 dominantly alters the channel gating of wild-type lens connexins.J Cell Sci. 2007 Dec 1;120(Pt 23):4107-16. doi: 10.1242/jcs.012237. Epub 2007 Nov 14. J Cell Sci. 2007. PMID: 18003700
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
-
Regulation of Connexin Gap Junctions and Hemichannels by Calcium and Calcium Binding Protein Calmodulin.Int J Mol Sci. 2020 Nov 2;21(21):8194. doi: 10.3390/ijms21218194. Int J Mol Sci. 2020. PMID: 33147690 Free PMC article. Review.
Cited by
-
Mix and match: investigating heteromeric and heterotypic gap junction channels in model systems and native tissues.FEBS Lett. 2014 Apr 17;588(8):1193-204. doi: 10.1016/j.febslet.2014.02.025. Epub 2014 Feb 20. FEBS Lett. 2014. PMID: 24561196 Free PMC article. Review.
-
A novel GJA3 mutation causing autosomal dominant congenital perinuclear cataracts.BMC Ophthalmol. 2025 Apr 2;25(1):164. doi: 10.1186/s12886-025-03978-0. BMC Ophthalmol. 2025. PMID: 40175916 Free PMC article.
-
Specificity of the connexin W3/4 locus for functional gap junction formation.Channels (Austin). 2016 Nov;10(6):453-65. doi: 10.1080/19336950.2016.1200775. Epub 2016 Jun 15. Channels (Austin). 2016. PMID: 27304225 Free PMC article.
-
Focus on lens connexins.BMC Cell Biol. 2017 Jan 17;18(Suppl 1):6. doi: 10.1186/s12860-016-0116-6. BMC Cell Biol. 2017. PMID: 28124626 Free PMC article. Review.
-
The Hydrophobic Residues in Amino Terminal Domains of Cx46 and Cx50 Are Important for Their Gap Junction Channel Ion Permeation and Gating.Int J Mol Sci. 2022 Oct 1;23(19):11605. doi: 10.3390/ijms231911605. Int J Mol Sci. 2022. PMID: 36232905 Free PMC article.
References
-
- Addison PK, Berry V, Holden KR, Espinal D, Rivera B, Su H, Srivastava AK, Bhattacharya SS. A novel mutation in the connexin 46 gene (GJA3) causes autosomal dominant zonular pulverulent cataract in a Hispanic family. Mol Vis 12: 791–795, 2006 - PubMed
-
- Arora A, Minogue PJ, Liu X, Reddy MA, Ainsworth JR, Bhattacharya SS, Webster AR, Hunt DM, Ebihara L, Moore AT, Beyer EC, Berthoud VM. A novel GJA8 mutation is associated with autosomal dominant lamellar pulverulent cataract: further evidence for gap junction dysfunction in human cataract. J Med Genet 43: e2, 2006 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

