Characterization of sacral interneurons that mediate activation of locomotor pattern generators by sacrocaudal afferent input
- PMID: 23303951
- PMCID: PMC6704898
- DOI: 10.1523/JNEUROSCI.4390-12.2013
Characterization of sacral interneurons that mediate activation of locomotor pattern generators by sacrocaudal afferent input
Abstract
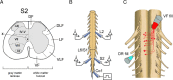
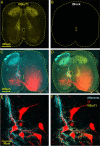
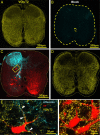
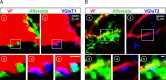
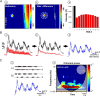
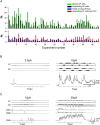
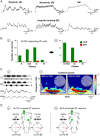
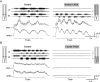
Identification of the neural pathways involved in retraining the spinal central pattern generators (CPGs) by afferent input in the absence of descending supraspinal control is feasible in isolated rodent spinal cords where the locomotor CPGs are potently activated by sacrocaudal afferent (SCA) input. Here we study the involvement of sacral neurons projecting rostrally through the ventral funiculi (VF) in activation of the CPGs by sensory stimulation. Fluorescent labeling and immunostaining showed that VF neurons are innervated by primary afferents immunoreactive for vesicular glutamate transporters 1 and 2 and by intraspinal neurons. Calcium imaging revealed that 55% of the VF neurons were activated by SCA stimulation. The activity of VF neurons and the sacral and lumbar CPGs was abolished when non-NMDA receptors in the sacral segments were blocked by the antagonist CNQX. When sacral NMDA receptors were blocked by APV, the sacral CPGs were suppressed, VF neurons with nonrhythmic activity were recruited and a moderate-drive locomotor rhythm developed during SCA stimulation. In contrast, when the sacral CPGs were activated by SCA stimulation, rhythmic and nonrhythmic VF neurons were recruited and the locomotor rhythm was most powerful. The activity of 73 and 27% of the rhythmic VF neurons was in-phase with the ipsilateral and contralateral motor output, respectively. Collectively, our studies indicate that sacral VF neurons serve as a major link between SCA and the hindlimb CPGs and that the ability of SCA to induce stepping can be enhanced by the sacral CPGs. The nature of the ascending drive to lumbar CPGs, the identity of subpopulations of VF neurons, and their potential role in activating the locomotor rhythm are discussed.
Figures


Similar articles
-
Shaping the Output of Lumbar Flexor Motoneurons by Sacral Neuronal Networks.J Neurosci. 2017 Feb 1;37(5):1294-1311. doi: 10.1523/JNEUROSCI.2213-16.2016. Epub 2016 Dec 26. J Neurosci. 2017. PMID: 28025254 Free PMC article.
-
Neuroanatomical basis for cholinergic modulation of locomotor networks by sacral relay neurons with ascending lumbar projections.J Comp Neurol. 2014 Oct 15;522(15):3437-55. doi: 10.1002/cne.23613. Epub 2014 May 5. J Comp Neurol. 2014. PMID: 24752570
-
Neural pathways between sacrocaudal afferents and lumbar pattern generators in neonatal rats.J Neurophysiol. 2003 Feb;89(2):773-84. doi: 10.1152/jn.00716.2002. J Neurophysiol. 2003. PMID: 12574455
-
The sacral networks and neural pathways used to elicit lumbar motor rhythm in the rodent spinal cord.Front Neural Circuits. 2014 Dec 3;8:143. doi: 10.3389/fncir.2014.00143. eCollection 2014. Front Neural Circuits. 2014. PMID: 25520624 Free PMC article. Review.
-
The motor output of hindlimb innervating segments of the spinal cord is modulated by cholinergic activation of rostrally projecting sacral relay neurons.J Mol Neurosci. 2014 Jul;53(3):517-24. doi: 10.1007/s12031-014-0351-2. Epub 2014 Jun 29. J Mol Neurosci. 2014. PMID: 24973872 Review.
Cited by
-
Spinal electrical stimulation to improve sympathetic autonomic functions needed for movement and exercise after spinal cord injury: a scoping clinical review.J Neurophysiol. 2022 Sep 1;128(3):649-670. doi: 10.1152/jn.00205.2022. Epub 2022 Jul 27. J Neurophysiol. 2022. PMID: 35894427 Free PMC article.
-
Bimodal Respiratory-Locomotor Neurons in the Neonatal Rat Spinal Cord.J Neurosci. 2016 Jan 20;36(3):926-37. doi: 10.1523/JNEUROSCI.1825-15.2016. J Neurosci. 2016. PMID: 26791221 Free PMC article.
-
Unique Spatiotemporal Neuromodulation of the Lumbosacral Circuitry Shapes Locomotor Success after Spinal Cord Injury.J Neurotrauma. 2016 Sep 15;33(18):1709-23. doi: 10.1089/neu.2015.4256. Epub 2016 Apr 20. J Neurotrauma. 2016. PMID: 26792233 Free PMC article.
-
Shaping the Output of Lumbar Flexor Motoneurons by Sacral Neuronal Networks.J Neurosci. 2017 Feb 1;37(5):1294-1311. doi: 10.1523/JNEUROSCI.2213-16.2016. Epub 2016 Dec 26. J Neurosci. 2017. PMID: 28025254 Free PMC article.
-
A New Measure for Quantifying Four-Limb Coordination of Human Gait Based on Mobility Sensors.Sensors (Basel). 2024 Sep 21;24(18):6105. doi: 10.3390/s24186105. Sensors (Basel). 2024. PMID: 39338850 Free PMC article.
References
-
- Alvarez FJ, Villalba RM, Zerda R, Schneider SP. Vesicular glutamate transporters in the spinal cord, with special reference to sensory primary afferent synapses. J Comp Neurol. 2004;472:257–280. - PubMed
-
- Arshavsky YI, Deliagina TG, Orlovsky GN. Pattern generation. Curr Opin Neurobiol. 1997;7:781–789. - PubMed
-
- Blivis D, Mentis GZ, O'Donovan MJ, Lev-Tov A. Differential effects of opioids on sacrocaudal afferent pathways and central pattern generators in the neonatal rat spinal cord. J Neurophysiol. 2007;97:2875–2886. - PubMed
-
- Bonnot A, Mentis GZ, Skoch J, O'Donovan MJ. Electroporation loading of calcium-sensitive dyes into the CNS. J Neurophysiol. 2005;93:1793–1808. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
