Organization of cortical and thalamic input to pyramidal neurons in mouse motor cortex
- PMID: 23303952
- PMCID: PMC3710148
- DOI: 10.1523/JNEUROSCI.4338-12.2013
Organization of cortical and thalamic input to pyramidal neurons in mouse motor cortex
Abstract
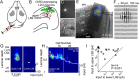
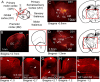
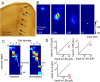
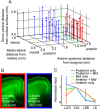
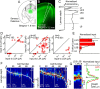
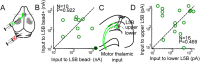
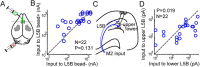
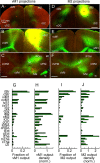
Determining how long-range synaptic inputs engage pyramidal neurons in primary motor cortex (M1) is important for understanding circuit mechanisms involved in regulating movement. We used channelrhodopsin-2-assisted circuit mapping to characterize the long-range excitatory synaptic connections made by multiple cortical and thalamic areas onto pyramidal neurons in mouse vibrissal motor cortex (vM1). Each projection innervated vM1 pyramidal neurons with a unique laminar profile. Collectively, the profiles for different sources of input partially overlapped and spanned all cortical layers. Specifically, orbital cortex (OC) inputs primarily targeted neurons in L6. Secondary motor cortex (M2) inputs excited neurons mainly in L5B, including pyramidal tract neurons. In contrast, thalamocortical inputs from anterior motor-related thalamic regions, including VA/VL (ventral anterior thalamic nucleus/ventrolateral thalamic nucleus), targeted neurons in L2/3 through L5B, but avoided L6. Inputs from posterior sensory-related thalamic areas, including POm (posterior thalamic nuclear group), targeted neurons only in the upper layers (L2/3 and L5A), similar to inputs from somatosensory (barrel) cortex. Our results show that long-range excitatory inputs target vM1 pyramidal neurons in a layer-specific manner. Inputs from sensory-related cortical and thalamic areas preferentially target the upper-layer pyramidal neurons in vM1. In contrast, inputs from OC and M2, areas associated with volitional and cognitive aspects of movements, bypass local circuitry and have direct monosynaptic access to neurons projecting to brainstem and thalamus.
Figures




Similar articles
-
Organization of Cortical and Thalamic Input to Inhibitory Neurons in Mouse Motor Cortex.J Neurosci. 2022 Oct 26;42(43):8095-8112. doi: 10.1523/JNEUROSCI.0950-22.2022. Epub 2022 Sep 14. J Neurosci. 2022. PMID: 36104281 Free PMC article.
-
Anterolateral Motor Cortex Connects with a Medial Subdivision of Ventromedial Thalamus through Cell Type-Specific Circuits, Forming an Excitatory Thalamo-Cortico-Thalamic Loop via Layer 1 Apical Tuft Dendrites of Layer 5B Pyramidal Tract Type Neurons.J Neurosci. 2018 Oct 10;38(41):8787-8797. doi: 10.1523/JNEUROSCI.1333-18.2018. Epub 2018 Aug 24. J Neurosci. 2018. PMID: 30143573 Free PMC article.
-
Synaptic circuit organization of motor corticothalamic neurons.J Neurosci. 2015 Feb 4;35(5):2293-307. doi: 10.1523/JNEUROSCI.4023-14.2015. J Neurosci. 2015. PMID: 25653383 Free PMC article.
-
Local connections of excitatory neurons in motor-associated cortical areas of the rat.Front Neural Circuits. 2013 May 28;7:75. doi: 10.3389/fncir.2013.00075. eCollection 2013. Front Neural Circuits. 2013. PMID: 23754982 Free PMC article. Review.
-
Input-output organization of the ventrolateral nucleus of the thalamus.Stereotact Funct Neurosurg. 1993;60(1-3):17-31. doi: 10.1159/000100587. Stereotact Funct Neurosurg. 1993. PMID: 8511431 Review.
Cited by
-
Multitarget Multiscale Simulation for Pharmacological Treatment of Dystonia in Motor Cortex.Front Pharmacol. 2016 Jun 14;7:157. doi: 10.3389/fphar.2016.00157. eCollection 2016. Front Pharmacol. 2016. PMID: 27378922 Free PMC article.
-
A Corticocortical Circuit Directly Links Retrosplenial Cortex to M2 in the Mouse.J Neurosci. 2016 Sep 7;36(36):9365-74. doi: 10.1523/JNEUROSCI.1099-16.2016. J Neurosci. 2016. PMID: 27605612 Free PMC article.
-
Correlated Somatosensory Input in Parvalbumin/Pyramidal Cells in Mouse Motor Cortex.eNeuro. 2023 May 8;10(5):ENEURO.0488-22.2023. doi: 10.1523/ENEURO.0488-22.2023. Print 2023 May. eNeuro. 2023. PMID: 37094939 Free PMC article.
-
Cortical depth-dependent human fMRI of resting-state networks using EPIK.Front Neurosci. 2023 May 18;17:1151544. doi: 10.3389/fnins.2023.1151544. eCollection 2023. Front Neurosci. 2023. PMID: 37274214 Free PMC article.
-
A genuine layer 4 in motor cortex with prototypical synaptic circuit connectivity.Elife. 2014 Dec 19;3:e05422. doi: 10.7554/eLife.05422. Elife. 2014. PMID: 25525751 Free PMC article.
References
-
- Asanuma H. Functional role of sensory inputs to the motor cortex. Prog Neurobiol. 1981;16:241–262. - PubMed
-
- Asanuma H, Larsen KD, Yumiya H. Receptive fields of thalamic neurons projecting to the motor cortex in the cat. Brain Res. 1979;172:217–228. - PubMed
-
- Asanuma H, Larsen K, Yumiya H. Peripheral input pathways to the monkey motor cortex. Exp Brain Res. 1980;38:349–355. - PubMed
-
- Brecht M. Movement, confusion, and orienting in frontal cortices. Neuron. 2011;72:193–196. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
