Proteomic analysis reveals the deregulation of inflammation-related proteins in acupuncture-treated rats with asthma onset
- PMID: 23304218
- PMCID: PMC3523810
- DOI: 10.1155/2012/850512
Proteomic analysis reveals the deregulation of inflammation-related proteins in acupuncture-treated rats with asthma onset
Abstract
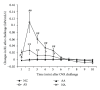
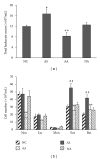
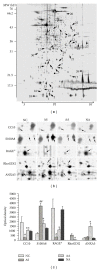
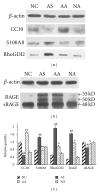
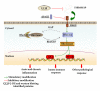
Although the beneficial effects of acupuncture in asthma treatment have been well documented, little is known regarding the biological basis of this treatment. Changes in the lung proteome of acupuncture-treated rats with asthma onset were comparatively analyzed using a two-dimensional gel electrophoresis (2DE) and mass-spectrometry- (MS-) based proteomic approach. Acupuncture on specific acupuncture points appeared to improve respiratory function and reduce the total number of leukocytes and eosinophils in bronchoalveolar lavage fluid in OVA-induced asthma onset. Image analysis of 2DE gels revealed 32 differentially expressed acupuncture-specific protein spots in asthma onset; 30 of which were successfully identified as 28 unique proteins using LC-MS/MS. Bioinformatic analyses indicated that these altered proteins are most likely involved in inflammation-related biological functions, and the functional associations of these proteins result in an inflammation signaling pathway. Acupuncture regulates the pathway at different levels by regulating several key nodal proteins, including downregulating of proinflammatory proteins (e.g., S100A8, RAGE, and S100A11) and upregulating of anti-inflammatory proteins (e.g., CC10, ANXA5, and sRAGE). These deregulated inflammation-related proteins may mediate, at least in part, the antiasthmatic effect of acupuncture. Further functional investigation of these acupuncture-specific effector proteins could identify new drug candidates for the prophylaxis and treatment of asthma.
Figures
Similar articles
-
Non-specific physiological background effects of acupuncture revealed by proteomic analysis in normal rats.BMC Complement Altern Med. 2014 Oct 5;14:375. doi: 10.1186/1472-6882-14-375. BMC Complement Altern Med. 2014. PMID: 25282142 Free PMC article.
-
TMT-based quantitative proteomics reveals suppression of SLC3A2 and ATP1A3 expression contributes to the inhibitory role of acupuncture on airway inflammation in an OVA-induced mouse asthma model.Biomed Pharmacother. 2021 Feb;134:111001. doi: 10.1016/j.biopha.2020.111001. Epub 2020 Dec 16. Biomed Pharmacother. 2021. PMID: 33341053
-
Acupuncture Attenuated Inflammation and Inhibited Th17 and Treg Activity in Experimental Asthma.Evid Based Complement Alternat Med. 2015;2015:340126. doi: 10.1155/2015/340126. Epub 2015 Nov 3. Evid Based Complement Alternat Med. 2015. PMID: 26612993 Free PMC article.
-
Acupuncture inhibited airway inflammation and group 2 innate lymphoid cells in the lung in an ovalbumin-induced murine asthma model.Acupunct Med. 2021 Jun;39(3):217-225. doi: 10.1177/0964528420924033. Epub 2020 Jun 15. Acupunct Med. 2021. PMID: 32539427
-
[The role of S100A8/RAGE and Caveolin-1 and the effect of roxithromycin on their expression in a rat model of neutrophilic asthma].Zhonghua Jie He He Hu Xi Za Zhi. 2019 Nov 12;42(11):845-851. doi: 10.3760/cma.j.issn.1001-0939.2019.11.012. Zhonghua Jie He He Hu Xi Za Zhi. 2019. PMID: 31694095 Chinese.
Cited by
-
Electroacupuncture inhibition of hyperalgesia in rats with adjuvant arthritis: involvement of cannabinoid receptor 1 and dopamine receptor subtypes in striatum.Evid Based Complement Alternat Med. 2013;2013:393460. doi: 10.1155/2013/393460. Epub 2013 May 25. Evid Based Complement Alternat Med. 2013. PMID: 23762129 Free PMC article.
-
Serum from acupuncture-treated asthmatic rats regulates p38-MAPK activation in airway smooth muscle cells.Am J Transl Res. 2023 Feb 15;15(2):1063-1071. eCollection 2023. Am J Transl Res. 2023. PMID: 36915784 Free PMC article.
-
Conventional Treatments plus Acupuncture for Asthma in Adults and Adolescent: A Systematic Review and Meta-Analysis.Evid Based Complement Alternat Med. 2019 Jan 17;2019:9580670. doi: 10.1155/2019/9580670. eCollection 2019. Evid Based Complement Alternat Med. 2019. PMID: 30792747 Free PMC article. Review.
-
Non-specific physiological background effects of acupuncture revealed by proteomic analysis in normal rats.BMC Complement Altern Med. 2014 Oct 5;14:375. doi: 10.1186/1472-6882-14-375. BMC Complement Altern Med. 2014. PMID: 25282142 Free PMC article.
-
Electroacupuncture Alleviates Diabetic Peripheral Neuropathy by Regulating Glycolipid-Related GLO/AGEs/RAGE Axis.Front Endocrinol (Lausanne). 2021 Jul 6;12:655591. doi: 10.3389/fendo.2021.655591. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34295304 Free PMC article.
References
-
- Global Initiative for Asthma (GINA) GINA Report, Global Strategy for Asthma Management and Prevention, 2010, http://www.ginasthma.org.
-
- Fang C, Corrigan CJ, Ying S. The treatment targets of asthma: from laboratory to clinic. Inflammation & Allergy Drug Targets. 2008;7(2):119–128. - PubMed
-
- Barnes PJ. New therapies for asthma: is there any progress? Trends in Pharmacological Sciences. 2010;31(7):335–343. - PubMed
-
- Marino LA, Shen J. Characteristics of complementary and alternative medicine use among adults with current asthma, 2006. Journal of Asthma. 2010;47(5):521–525. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

