Intracellular NAD(H) levels control motility and invasion of glioma cells
- PMID: 23307072
- PMCID: PMC11113314
- DOI: 10.1007/s00018-012-1249-1
Intracellular NAD(H) levels control motility and invasion of glioma cells
Abstract
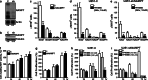
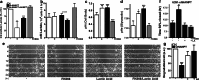
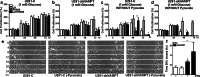
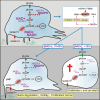
Oncogenic transformation involves reprogramming of cell metabolism, whereby steady-state levels of intracellular NAD(+) and NADH can undergo dramatic changes while ATP concentration is generally well maintained. Altered expression of nicotinamide phosphoribosyltransferase (NAMPT), the rate-limiting enzyme of NAD(+)-salvage, accompanies the changes in NAD(H) during tumorigenesis. Here, we show by genetic and pharmacological inhibition of NAMPT in glioma cells that fluctuation in intracellular [NAD(H)] differentially affects cell growth and morphodynamics, with motility/invasion capacity showing the highest sensitivity to [NAD(H)] decrease. Extracellular supplementation of NAD(+) or re-expression of NAMPT abolished the effects. The effects of NAD(H) decrease on cell motility appeared parallel coupled with diminished pyruvate-lactate conversion by lactate dehydrogenase (LDH) and with changes in intracellular and extracellular pH. The addition of lactic acid rescued and knockdown of LDH-A replicated the effects of [NAD(H)] on motility. Combined, our observations demonstrate that [NAD(H)] is an important metabolic component of cancer cell motility. Nutrient or drug-mediated modulation of NAD(H) levels may therefore represent a new option for blocking the invasive behavior of tumors.
Figures





References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

