Highly fluorescent semiconducting polymer dots for biology and medicine
- PMID: 23307291
- PMCID: PMC5616106
- DOI: 10.1002/anie.201205133
Highly fluorescent semiconducting polymer dots for biology and medicine
Abstract
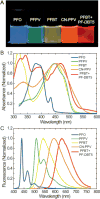
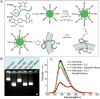
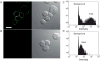
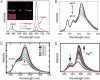
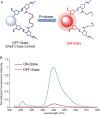
In recent years, semiconducting polymer nanoparticles have attracted considerable attention because of their outstanding characteristics as fluorescent probes. These nanoparticles, which primarily consist of π-conjugated polymers and are called polymer dots (Pdots) when they exhibit small particle size and high brightness, have demonstrated utility in a wide range of applications such as fluorescence imaging and biosensing. In this review, we summarize recent findings of the photophysical properties of Pdots which speak to the merits of these entities as fluorescent labels. This review also highlights the surface functionalization and biomolecular conjugation of Pdots, and their applications in cellular labeling, in vivo imaging, single-particle tracking, biosensing, and drug delivery. We discuss the relationship between the physical properties and performance, and evaluate the merits and limitations of the Pdot probes for certain imaging tasks and fluorescence assays. We also tackle the current challenges of Pdots and share our perspective on the future directions of the field.
Copyright © 2013 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures











References
-
- Evanko D. Nat Methods. 2005;2:901. This issue includes a series of review articles on fluorescence imaging techniques. - PubMed
-
- Pepperkok R, Ellenberg J. Nat Rev Mol Cell Biol. 2006;7:690. - PubMed
-
- Lakowicz JR. Principles of Fluorescence Spectroscopy. 3rd. Springer; 2006.
-
- Mason WT. Fluorescent and Luminescent Probes for Biological Activity. 2nd. Academic Press; London: 1999.
-
- Yildiz A, Forkey JN, McKinney SA, Ha T, Goldman YE, Selvin PR. Science. 2003;300:2061. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

