Capture of uropathogenic E. coli by using synthetic glycan ligands specific for the pap-pilus
- PMID: 23307594
- PMCID: PMC5453672
- DOI: 10.1002/cbic.201200582
Capture of uropathogenic E. coli by using synthetic glycan ligands specific for the pap-pilus
Abstract
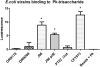
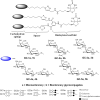
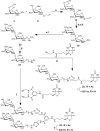
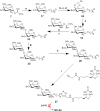
Biotinylated mono- and biantennary di-/trisaccharides were synthesized to evaluate their ability to capture E. coli strains that express pilus types with different receptor specificities. The synthesized biotinylated di-/trisaccharides contain Galα(1→4)Gal, Galα(1→4)GalNHAc, GalNHAcα(1→4)Gal, Galα(1→4)Galβ(1→4)Glc and GalNHAcα(1→4)Galβ(1→4)Glc as carbohydrate epitopes. These biotinylated oligosaccharides were immobilized on streptavidin-coated magnetic beads, and incubated with different strains of live E. coli. Capturing ability was assessed by using a luciferase assay that detects bacterial ATP. The trisaccharides containing Galα(1→4)Galβ(1→4)Glc and the disaccharides containing Galα(1→4)Gal as the epitopes exhibited strong capturing ability for uropathogenic E. coli strains with the pap pilus genotype, including CFT073, J96 and J96 pilE. The same ligands failed to capture E. coli strains with fim, prs, or foc genotypes. Uropathogenic CFT073 was also captured moderately by biantennary disaccharides containing a GalNHAc moiety at the reducing end; however, other saccharides containing GalNHAc at the nonreducing end did not capture the CFT073 strain. These synthetic glycoconjugates could potentially be adapted as rapid diagnostic agents to differentiate between different E. coli pathovars.
Copyright © 2013 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures

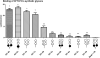

Similar articles
-
Isogenic P-fimbrial deletion mutants of pyelonephritogenic Escherichia coli: the role of alpha Gal(1-4) beta Gal binding in virulence of a wild-type strain.Mol Microbiol. 1993 Oct;10(1):143-55. doi: 10.1111/j.1365-2958.1993.tb00911.x. Mol Microbiol. 1993. PMID: 7968511
-
The design and synthesis of an α-Gal trisaccharide epitope that provides a highly specific anti-Gal immune response.Org Biomol Chem. 2017 Apr 5;15(14):2979-2992. doi: 10.1039/c7ob00448f. Org Biomol Chem. 2017. PMID: 28294277 Free PMC article.
-
Parasite glycoconjugates. Part 16: Synthesis of a disaccharide and phosphorylated di- and tri-saccharides from Leishmania lipophosphoglycan.Carbohydr Res. 2006 Aug 14;341(11):1954-64. doi: 10.1016/j.carres.2006.03.026. Epub 2006 May 15. Carbohydr Res. 2006. PMID: 16697981
-
Regulation of type 1 fimbriae by unlinked FimB- and FimE-like recombinases in uropathogenic Escherichia coli strain CFT073.Infect Immun. 2006 Feb;74(2):1072-83. doi: 10.1128/IAI.74.2.1072-1083.2006. Infect Immun. 2006. PMID: 16428754 Free PMC article.
-
Expression of binding properties of Gal/GalNAc reactive lectins by mammalian glycotopes (an updated report).Adv Exp Med Biol. 2001;491:55-64. doi: 10.1007/978-1-4615-1267-7_4. Adv Exp Med Biol. 2001. PMID: 14533789 Review.
Cited by
-
Binding of Pk-trisaccharide analogs of globotriaosylceramide to Shiga toxin variants.Infect Immun. 2013 Aug;81(8):2753-60. doi: 10.1128/IAI.00274-13. Epub 2013 May 20. Infect Immun. 2013. PMID: 23690406 Free PMC article.
-
Rapid Isolation of Low-Level Carbapenem-Resistant E. coli from Water and Foods Using Glycan-Coated Magnetic Nanoparticles.Biosensors (Basel). 2023 Sep 23;13(10):902. doi: 10.3390/bios13100902. Biosensors (Basel). 2023. PMID: 37887095 Free PMC article.
-
Gold Nanoparticle-Based Plasmonic Detection of Escherichia coli, Salmonella enterica, Campylobacter jejuni, and Listeria monocytogenes from Bovine Fecal Samples.Microorganisms. 2024 May 25;12(6):1069. doi: 10.3390/microorganisms12061069. Microorganisms. 2024. PMID: 38930450 Free PMC article.
-
Current Methods for Extraction and Concentration of Foodborne Bacteria with Glycan-Coated Magnetic Nanoparticles: A Review.Biosensors (Basel). 2022 Feb 11;12(2):112. doi: 10.3390/bios12020112. Biosensors (Basel). 2022. PMID: 35200372 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

