Prediction and analysis of antibody amyloidogenesis from sequences
- PMID: 23308169
- PMCID: PMC3538782
- DOI: 10.1371/journal.pone.0053235
Prediction and analysis of antibody amyloidogenesis from sequences
Abstract
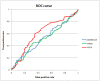
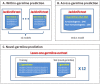
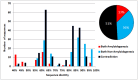
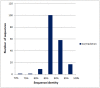
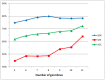
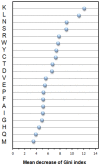
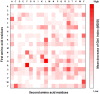
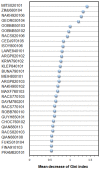
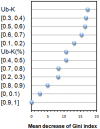
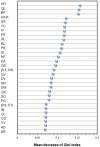
Antibody amyloidogenesis is the aggregation of soluble proteins into amyloid fibrils that is one of major causes of the failures of humanized antibodies. The prediction and prevention of antibody amyloidogenesis are helpful for restoring and enhancing therapeutic effects. Due to a large number of possible germlines, the existing method is not practical to predict sequences of novel germlines, which establishes individual models for each known germline. This study proposes a first automatic and across-germline prediction method (named AbAmyloid) capable of predicting antibody amyloidogenesis from sequences. Since the amyloidogenesis is determined by a whole sequence of an antibody rather than germline-dependent properties such as mutated residues, this study assess three types of germline-independent sequence features (amino acid composition, dipeptide composition and physicochemical properties). AbAmyloid using a Random Forests classifier with dipeptide composition performs well on a data set of 12 germlines. The within- and across-germline prediction accuracies are 83.10% and 83.33% using Jackknife tests, respectively, and the novel-germline prediction accuracy using a leave-one-germline-out test is 72.22%. A thorough analysis of sequence features is conducted to identify informative properties for further providing insights to antibody amyloidogenesis. Some identified informative physicochemical properties are amphiphilicity, hydrophobicity, reverse turn, helical structure, isoelectric point, net charge, mutability, coil, turn, linker, nuclear protein, etc. Additionally, the numbers of ubiquitylation sites in amyloidogenic and non-amyloidogenic antibodies are found to be significantly different. It reveals that antibodies less likely to be ubiquitylated tend to be amyloidogenic. The method AbAmyloid capable of automatically predicting antibody amyloidogenesis of novel germlines is implemented as a publicly available web server at http://iclab.life.nctu.edu.tw/abamyloid.
Conflict of interest statement
Figures

Similar articles
-
Computational identification of ubiquitylation sites from protein sequences.BMC Bioinformatics. 2008 Jul 15;9:310. doi: 10.1186/1471-2105-9-310. BMC Bioinformatics. 2008. PMID: 18625080 Free PMC article.
-
Using simple artificial intelligence methods for predicting amyloidogenesis in antibodies.BMC Bioinformatics. 2010 Feb 8;11:79. doi: 10.1186/1471-2105-11-79. BMC Bioinformatics. 2010. PMID: 20144194 Free PMC article.
-
Prediction and analysis of protein solubility using a novel scoring card method with dipeptide composition.BMC Bioinformatics. 2012;13 Suppl 17(Suppl 17):S3. doi: 10.1186/1471-2105-13-S17-S3. Epub 2012 Dec 13. BMC Bioinformatics. 2012. PMID: 23282103 Free PMC article.
-
FISH Amyloid - a new method for finding amyloidogenic segments in proteins based on site specific co-occurrence of aminoacids.BMC Bioinformatics. 2014 Feb 24;15:54. doi: 10.1186/1471-2105-15-54. BMC Bioinformatics. 2014. PMID: 24564523 Free PMC article.
-
Computational Approaches to Identification of Aggregation Sites and the Mechanism of Amyloid Growth.Adv Exp Med Biol. 2015;855:213-39. doi: 10.1007/978-3-319-17344-3_9. Adv Exp Med Biol. 2015. PMID: 26149932 Review.
Cited by
-
Stability of Protein Pharmaceuticals: Recent Advances.Pharm Res. 2024 Jul;41(7):1301-1367. doi: 10.1007/s11095-024-03726-x. Epub 2024 Jun 27. Pharm Res. 2024. PMID: 38937372 Review.
-
Exploring the sequence features determining amyloidosis in human antibody light chains.Sci Rep. 2021 Jul 2;11(1):13785. doi: 10.1038/s41598-021-93019-9. Sci Rep. 2021. PMID: 34215782 Free PMC article.
-
AbAgIntPre: A deep learning method for predicting antibody-antigen interactions based on sequence information.Front Immunol. 2022 Dec 22;13:1053617. doi: 10.3389/fimmu.2022.1053617. eCollection 2022. Front Immunol. 2022. PMID: 36618397 Free PMC article.
-
Investigating Local Sequence-Structural Attributes of Amyloidogenic Light Chain Variable Domains.Proteins. 2025 Sep;93(9):1451-1464. doi: 10.1002/prot.26815. Epub 2025 Mar 4. Proteins. 2025. PMID: 40034034 Free PMC article.
-
Advanced computational approaches to understand protein aggregation.Biophys Rev (Melville). 2024 Apr 24;5(2):021302. doi: 10.1063/5.0180691. eCollection 2024 Jun. Biophys Rev (Melville). 2024. PMID: 38681860 Free PMC article. Review.
References
-
- Presta LG (2008) Molecular engineering and design of therapeutic antibodies. Curr Opin Immunol 20: 460–470. - PubMed
-
- Padlan E (1991) A possible procedure for reducing the immunogenicity of antibody variable domains while preserving their ligand-binding properties. Molecular Immunology 28: 489–498. - PubMed
-
- Clark M (2000) Antibody humanization: a case of the 'Emperor's new clothes'? Immunol Today 21: 397–402. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

